Comprehensive research-only screening solutions for multiple disease markers available worldwide.
A Myriad of Possibilities
Designed for researchers, the Miriad® product line delivers versatile, Research Use Only solutions for multi-marker screening across blood banks, tissue and eye banks, organ donor programs, and scientific studies. Available worldwide without regulatory barriers, Miriad® offers ready-made and tailored combinations for detecting multiple disease markers. Built on our RVF Technology™, these rapid antibody tests provide immediate, easy-to-interpret results that enhance research and support informed decision-making in donor evaluation and advanced study environments.
-
Immediate results
No waiting time, results do not vanish.
-
Reliable accuracy
Achieves high testing performance across multiple specimen matrices.
-
Multiplex testing
Multiple diseases tested on a single cartridge using one specimen.
-
No equipment or refrigeration required
Stores at room temperature (2–30 °C / 35–86 °F) with an 18-month shelf life.
-
Easy to use and interpret
Simple procedure with instant, easy-to-read results, no special training required.
-
For Research Use Only
Works with serum, plasma, and venipuncture blood.
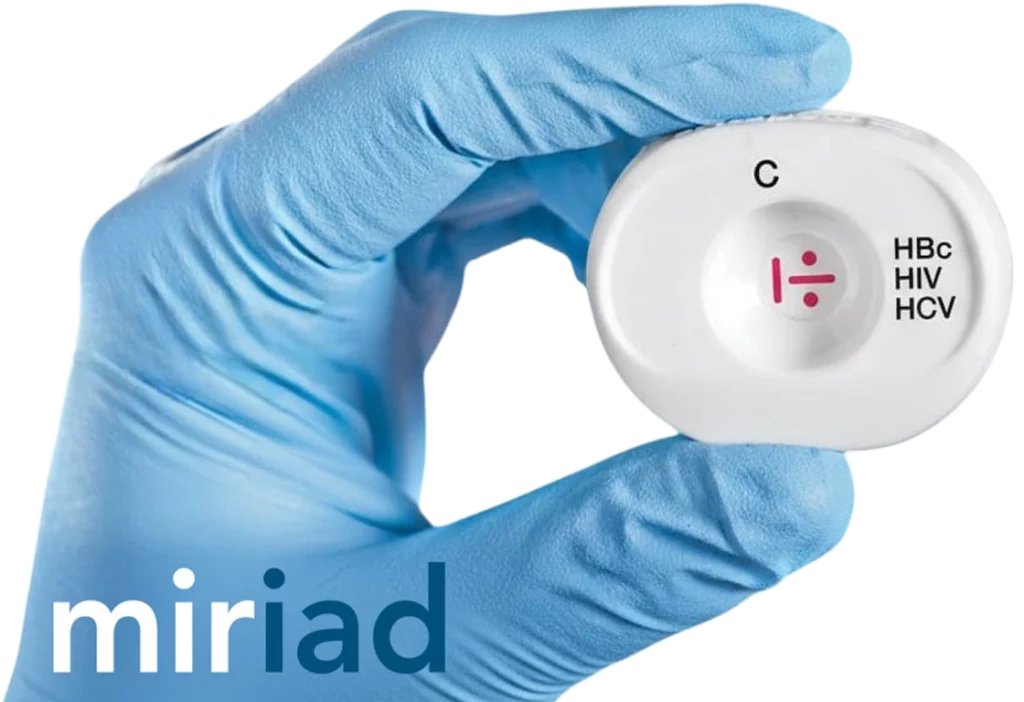
An essential tool for Tissue and Eye banks
Tissue and eye banks are using Miriad® as part of their initial donor screening, achieving a relative discard reduction of 40%. With Miriad®, tissue and eye banks add an immediate layer of actionable information to their procurement process, enabling valuable benefits such as:
-
Enhanced Technician Safety
Minimize exposure to sharps and infectious materials by reducing unnecessary handling of unsuitable tissue.
-
Reduced Operational Costs
Lower expenses for transportation, storage, and disposal by identifying ineligible tissue earlier in the process.
-
Less Stress for Donor Families
Faster screening enables quicker decisions, reducing the emotional burden on families during a sensitive time.
The Miriad® triple product has helped our organization perform preliminary screening of HIV and hepatitis. We are using these results to make initial determination of eligibility for donation. It also has proven to be beneficial as a safety measure for our technicians.
Kristen McCoy
VP Innovations, Eversight Illinois
to your needs
Miriad® rapid tests are trusted worldwide for use in both point-of-use (POU+) and laboratory (LAB+) settings. Through our distribution partners VWR, Medline and McKesson, we provide access to these formats designed for research use only, as well as test controls for selected tests to ensure consistent performance. Contact us for guidance on ordering process either direct or through our distributors.

- Qty. 25 tests
- Qty. 50 tests

- Cat.No. 815311006050
- Qty. 25 tests
- Downloads. Package Insert – EN
- Qty. 50 tests

- Qty. 25 tests
- Qty. 50 tests
- Cat.No. 815311006395
- Qty. 25 tests
- Cat.No. 815311006388
- Qty. 50 tests
- Cat.No. 815311005992
- VWR 77996-761
- Medline EDM311005992
- Qty. 25 tests
- Downloads Package Insert – EN
- Cat.No. 815311005961
- VWR 77996-759
- Qty. 50 tests
- Downloads Package Insert – EN
- Cat.No. 815311006012
- VWR 75780-214
- Medline EDM311006012
- Qty. 25 tests
- Downloads Package Insert – EN
- Cat.No. 815311005954
- VWR 75780-216
- Medline EDM311005954
- Qty. 50 tests
- Downloads Package Insert – EN

- Cat.No. 815311006005
- VWR 10789-423
- Medline EDM311006005
- Qty. 25 tests
- Downloads Package Insert – EN; Quick Reference Guide – EN
- Bulk Format
Cat. No. 815311006029
Qty. 400 tests
- Cat.No. 815311005978
- VWR 10847-590
- Medline EDM311005978
- Qty. 50 tests
- Downloads Package Insert – EN
- Cat.No. 815311006043
- Qty. 25 tests
- Downloads Package Insert – EN
- Qty. 50 tests

- Qty. 25 tests
- Qty. 50 tests
For customized combinations targeting multiple disease markers, our Miriad® RVF Toolkit provides a flexible, scalable solution designed to support a wide range of screening strategies.
Downloads and Links
Additional materials are available through your partner login or upon request.
Related Products
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Learn more about our upcoming technology and products advancing through our development pipeline.
Frequently Asked Questions
To learn more about the Miriad® product line, please contact us.
The Miriad® RVF Toolkit contains all essential materials for creating rapid analytical tools for antibody or antigen detection using MedMira’s patented Rapid Vertical Flow Technology™. It allows researchers to efficiently develop prototypes and explore new assay concepts for research-use applications.
Learn more
The Miriad® product line represents MedMira’s portfolio of research-use-only (RUO) rapid tests. These products require no regulatory approvals and can be sold globally for research purposes, making our RVF technology™ accessible to researchers, academics, blood banks, and other scientific institutions.
Miriad® tests are typically used at the earliest screening stage to provide rapid, actionable information before samples enter downstream laboratory testing. This helps reduce unnecessary processing of ineligible tissue and improves operational efficiency.
RVF™ (Rapid Vertical Flow™) is a flow-through technology that captures specific biomarkers from blood, serum, or plasma samples on a specialized membrane, delivering instant visual results.
Learn more
Rapid tests provide valuable advantages across a range of environments, from point-of-care settings to research and laboratory workflows. They deliver results within minutes without relying on extensive infrastructure, specialized instruments, or highly trained personnel, which helps reduce costs and streamline testing processes. Modern rapid assays often demonstrate performance comparable to laboratory methods, making them a practical option for decentralized screening, high-throughput research studies, emergency situations, and programs that require immediate decision-making or broad accessibility.



















