A platform for advanced multi-target testing with independent antigen and antibody detection enabling interference-free results.
One Platform. Any Combination.
The MEDDUO™ platform is designed to generate multiple results within the same device. It supports defined combinations of antibody and antigen targets as well as multiplex detection of up to eight (8) antibody or antigen analytes within one assay. The platform incorporates a dual-window read-out design that physically separates antigen and antibody detection pathways. This separation enables independent assay workflows, supports clear result interpretation, and prevents cross-interference between antigen- and antibody-based assays.
-
Immediate results
No waiting time, results do not vanish.
-
Reliable accuracy
Achieves high TP testing performance across multiple specimen matrices.
-
Antigen and antibody detection
Dual-chamber design enables interference-free antigen and antibody results.
-
No equipment or refrigeration required
Stores at room temperature (2–30 °C / 35–86 °F) with a 12-month shelf life.
-
Easy to use and interpret
Simple procedure with instant, easy-to-read results, no special training required.
-
Ideal for any setting
Works with serum, plasma, and whole blood (venipuncture or fingerstick)

Dual-Window Formats
In contrast to conventional single-window RVF cartridges, the MEDDUO™ platform utilizes differentiated buffer volumes, multiple reagent vials, and two independent read-out windows. This architecture enables flexible assay configuration while maintaining separation of detection chemistries. The two read-out windows can be configured in different modes:

both windows configured for antibody detection

both windows configured for antigen detection


both windows configured for antigen detection
in three simple Steps
Testing doesn’t have to be complicated. Our intuitive three-step process ensures instant, consistent results with minimal effort. In this example, the right read-out window is configured for antigen detection and the left read-out window for antibody detection.
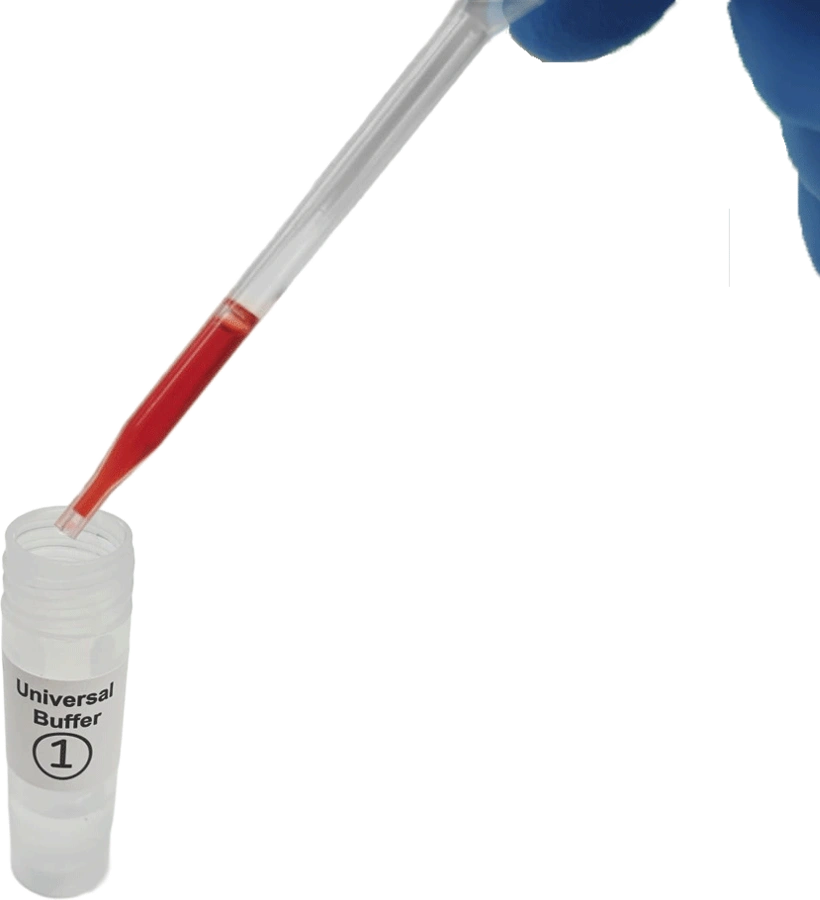
Add whole blood specimen to buffer in vial 1. Perform this step once only.
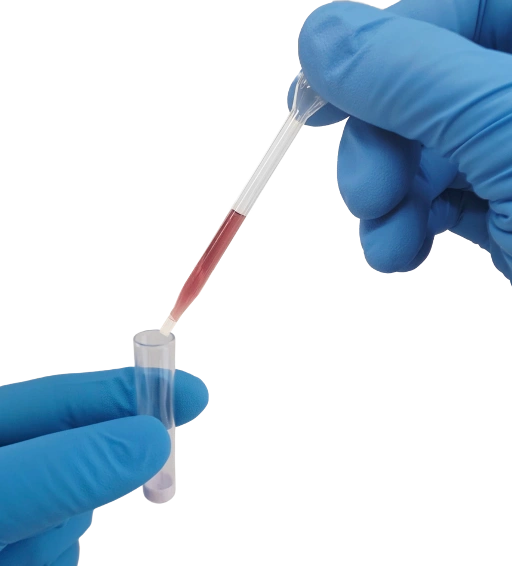
Using the supplied disposable pipette, add 10 drops from Step 1 to the lyophilize vial, mix to dissolve.
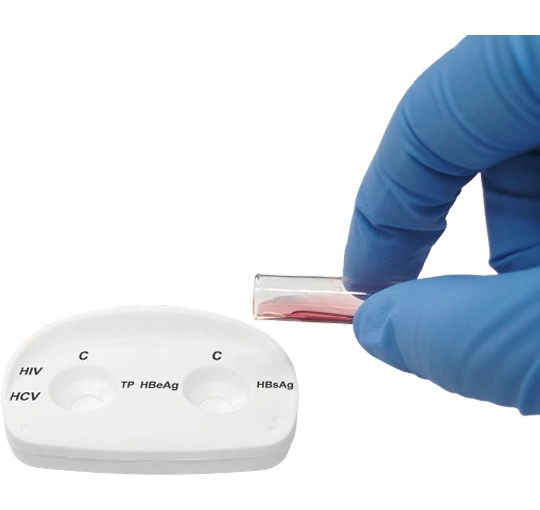
Pour the contents of the lyophilized vial onto the right reaction window.
Add whole blood specimen to buffer in vial 1. Perform this step once only.
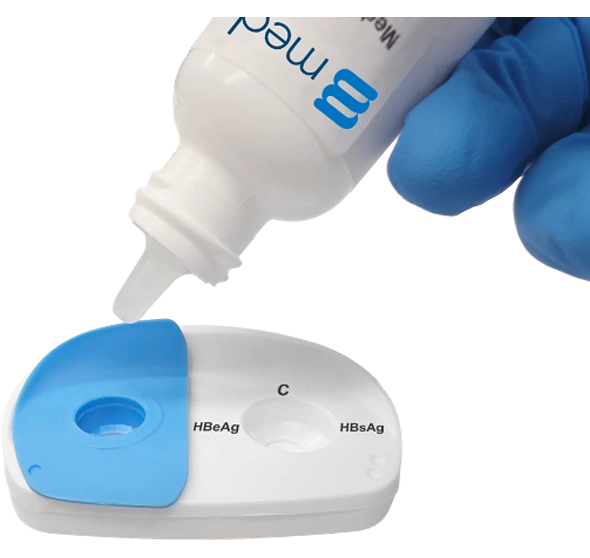
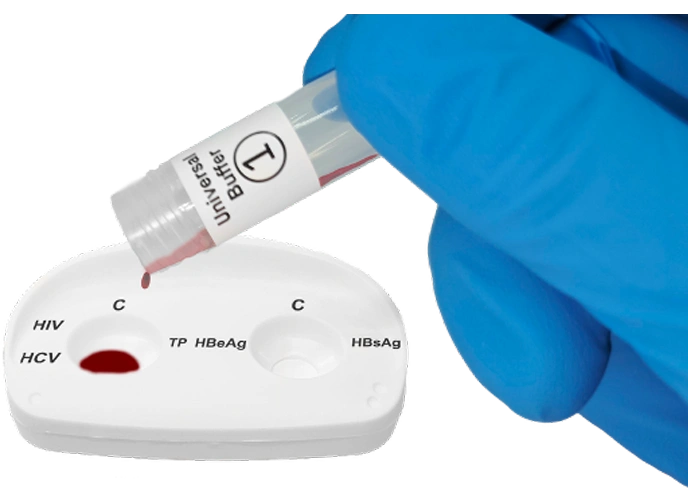
Place the InstantGold™ cap on the left reaction window and pour buffer onto it.
For a similar procedure, watch the Miriad® multiLinx™ HIV/HCV/TP Antibody & HBsAg/HBeAg Antigen video (Youtube, 5:05 min).
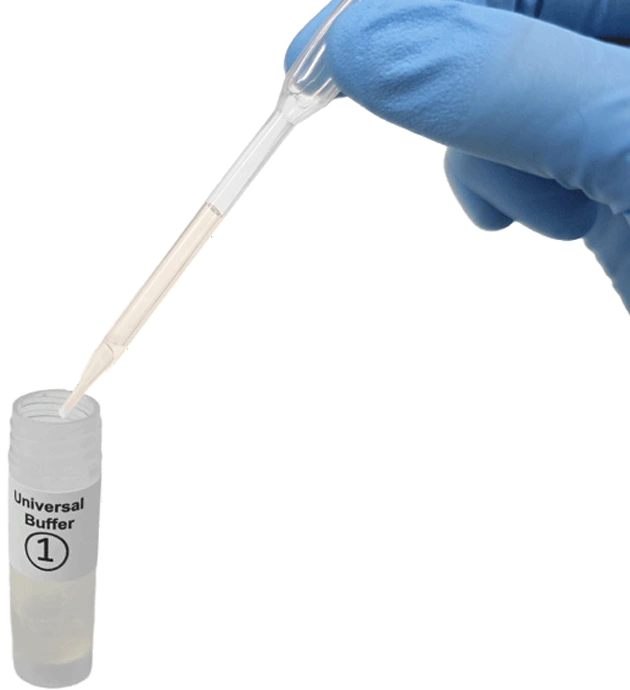
Add serum, plasma specimen to buffer in vial 1. Perform this step once only.
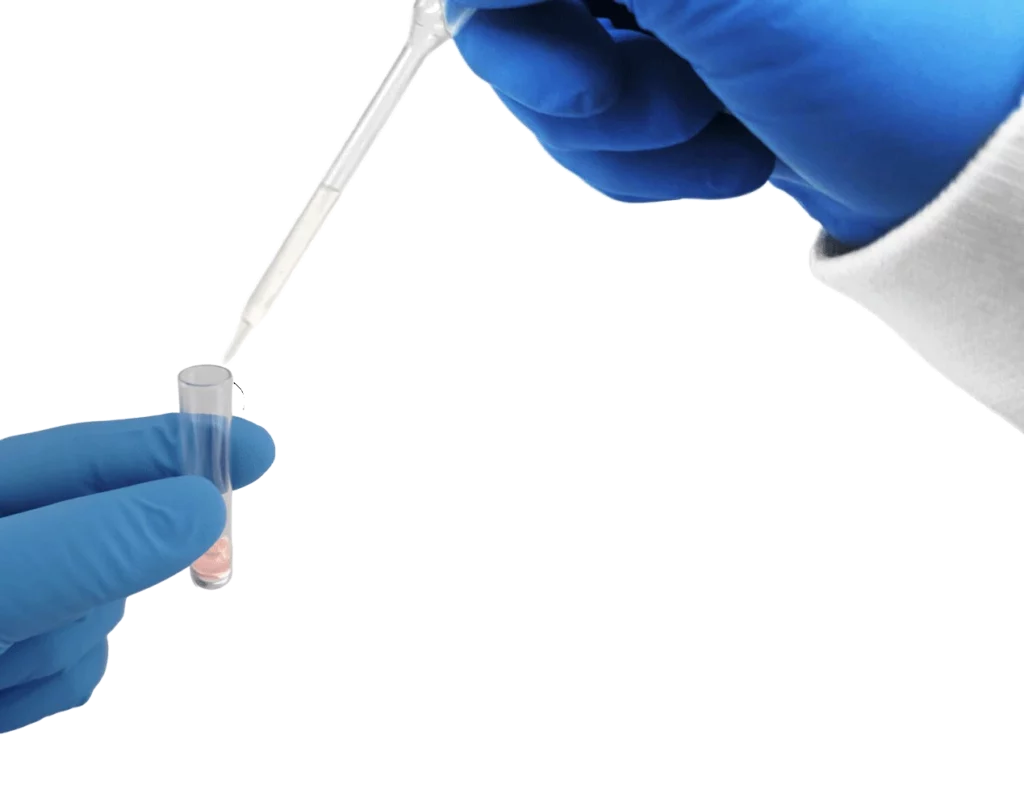
Add specimen and mix to reconstitute the lyophilized material until fully dissolved.

Pour the contents of the lyophilized vial onto the right reaction window.
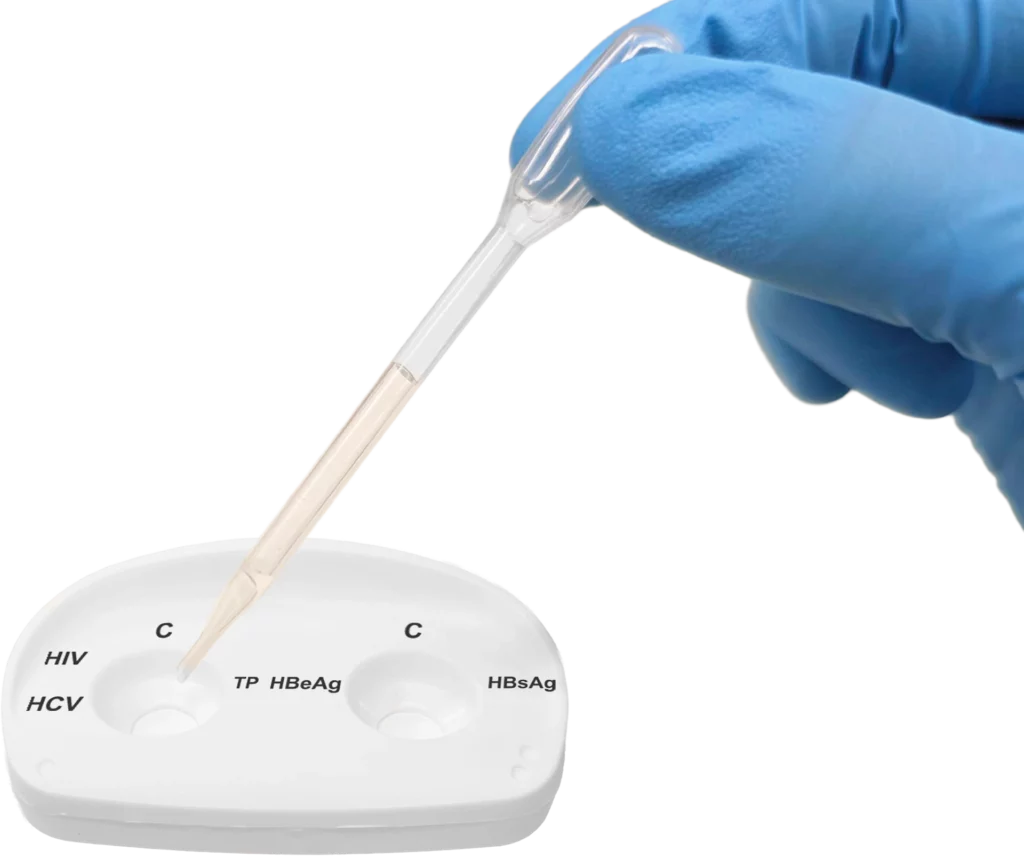
Pour the remaining specimen from vial 1 onto the left reaction window.

Place the InstantGold™ cap on the left reaction window and pour buffer onto it.
For a similar procedure, watch the Miriad® multiLinx™ HIV/HCV/TP Antibody & HBsAg/HBeAg Antigen video (Youtube, 5:05 min).
Explore your options with us
Interested in learning more about the MEDDUO™ platform, developing a custom assay, or exploring new testing concepts? We welcome your inquiry. Please contact us for further information.
Frequently Asked Questions
Dual-chamber technology separates antigen and antibody reactions into two independent test areas, preventing them from affecting each other. In some single-chamber assays that use biotin-streptavidin binding chemistry, elevated biotin (vitamin B7) levels in patient samples can disrupt assay performance and lead to unclear or inaccurate results. By physically separating antigen and antibody detection into independent chambers, MEDDUO™ reduces the risk of such interference and helps ensure clear, reliable, and interpretable results for combination antigen/antibody testing.
Multiplo® provides a single integrated readout for multi-parameter infection screening, ideal for broad public health testing. MEDDUO™, by contrast, uses a dual-window system that separates antigen and antibody detection, providing clear results without cross-interference and making it the preferred platform for next-generation antigen/antibody combination assays.
The MEDDUO™ platform is MedMira’s dual-window RVF cartridge architecture. MultiLinx™ assays represent multiplex test configurations that may be implemented on MEDDUO™ or standard RVF cartridges. Certain RUO assays such as HIV-HCV-TP-HBsAg-HBeAg distributed through partners are based on licensed MedMira technology and represent earlier-generation implementations.
No. The MEDDUO™ platform is designed as a configurable system that supports a wide range of antibody and antigen combinations. Assay configurations can be adapted based on target selection and application requirements. For further information or specific assay inquiries, please contact us.
Contact us
RVF (Rapid Vertical Flow) is a flow-through technology that captures specific biomarkers from blood, serum, or plasma samples on a specialized membrane, delivering instant visual results.
Learn more



















