
Accurate, rapid simultaneous detection of antibodies to HIV-1/2 and/or syphilis in whole blood, serum, and plasma specimens.
one test - two results
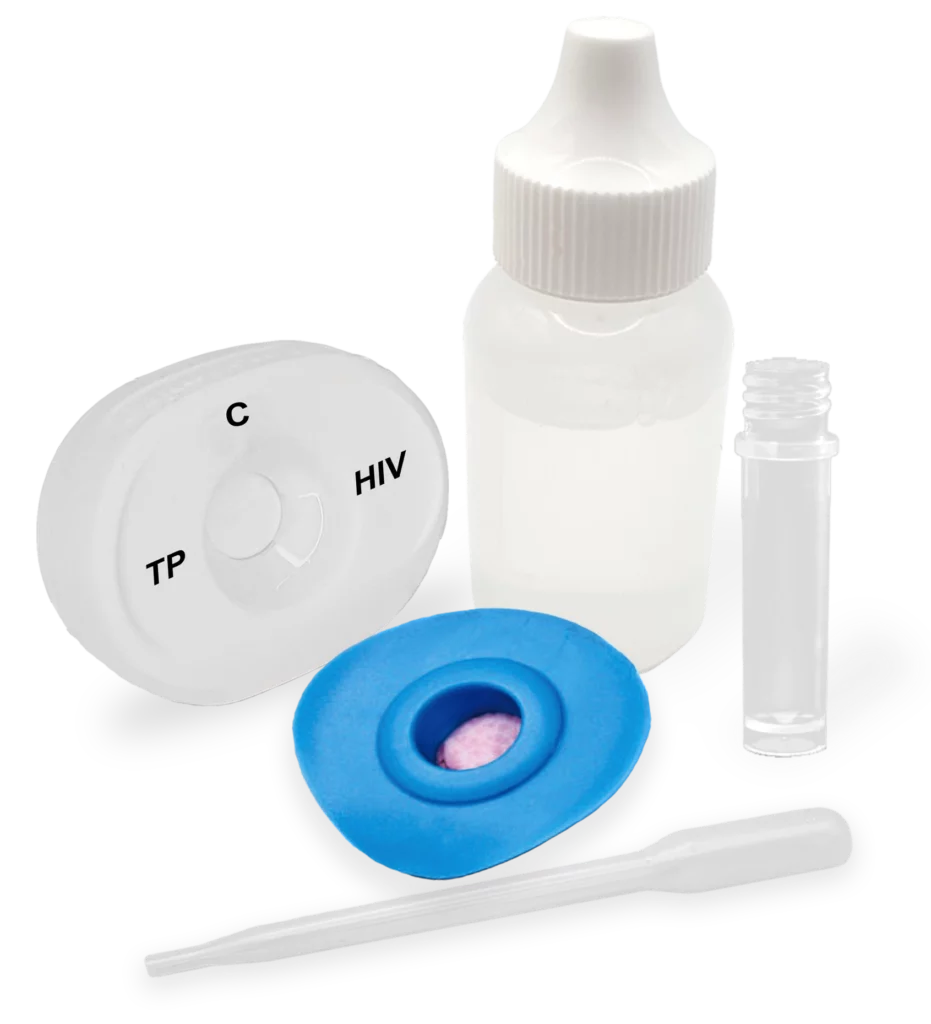
The Multiplo® Rapid TP/HIV Test enables simultaneous detection of antibodies to Treponema pallidum (syphilis) and HIV-1/2 in a single test using MedMira’s Rapid Vertical Flow (RVF) Technology™. Increasing co-infections – driven by shared risk factors and transmission routes – necessitate combined screening to support effective patient management and reduce mother-to-child transmission. Health Canada approved, our test provides fast, reliable point-of-care results from a single blood sample in just three simple steps. Its strong clinical performance supports timely decision-making, improving patient outcomes and contributing to broader public health goals.
-
Immediate results
No waiting time, results do not vanish.
-
Reliable accuracy
Achieves high testing performance.
-
Multiplex testing
Multiple diseases tested on a single cartridge using one blood sample.
-
No equipment or refrigeration required
Stores at room temperature (2–30 °C / 35–86 °F) with a 12-month shelf life.
-
Easy to use and interpret
Simple procedure with instant, easy-to-read results, no special training required.
-
Ideal for POC setting
Works with whole blood (fingerstick).

in three simple Steps
Testing doesn’t have to be complicated. Our intuitive three-step process ensures instant, consistent results with minimal effort.
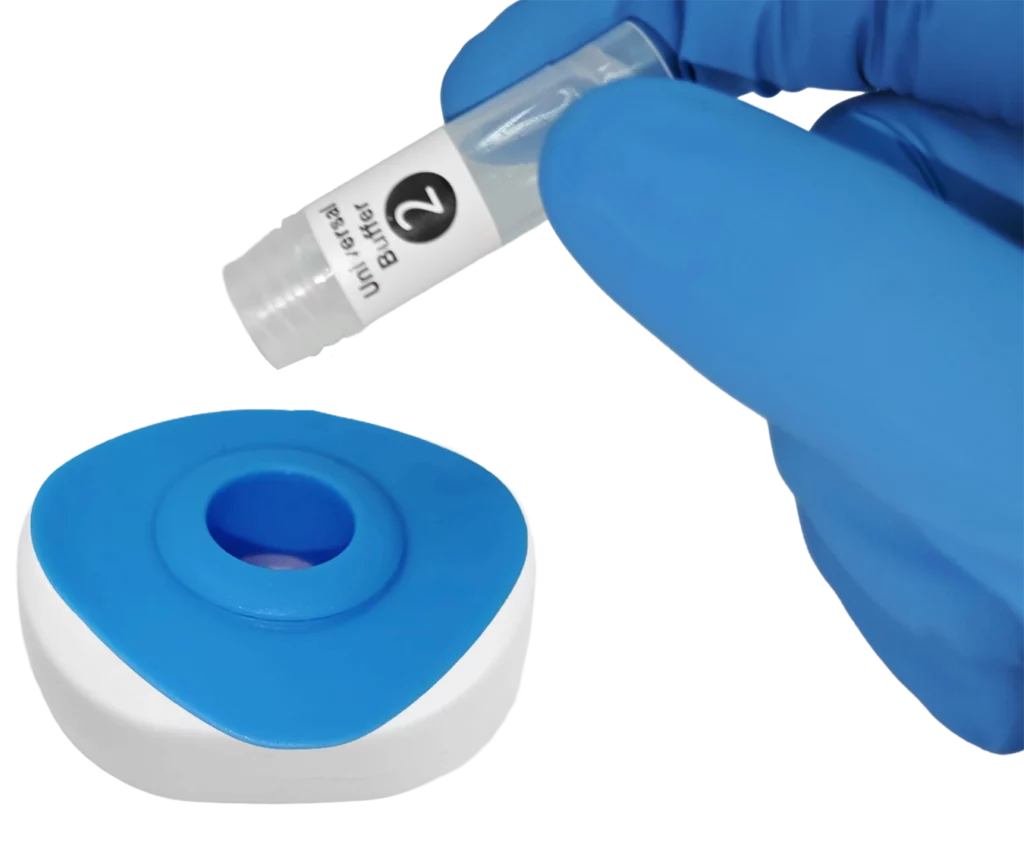
Pour buffer onto the InstantGold™ cap.
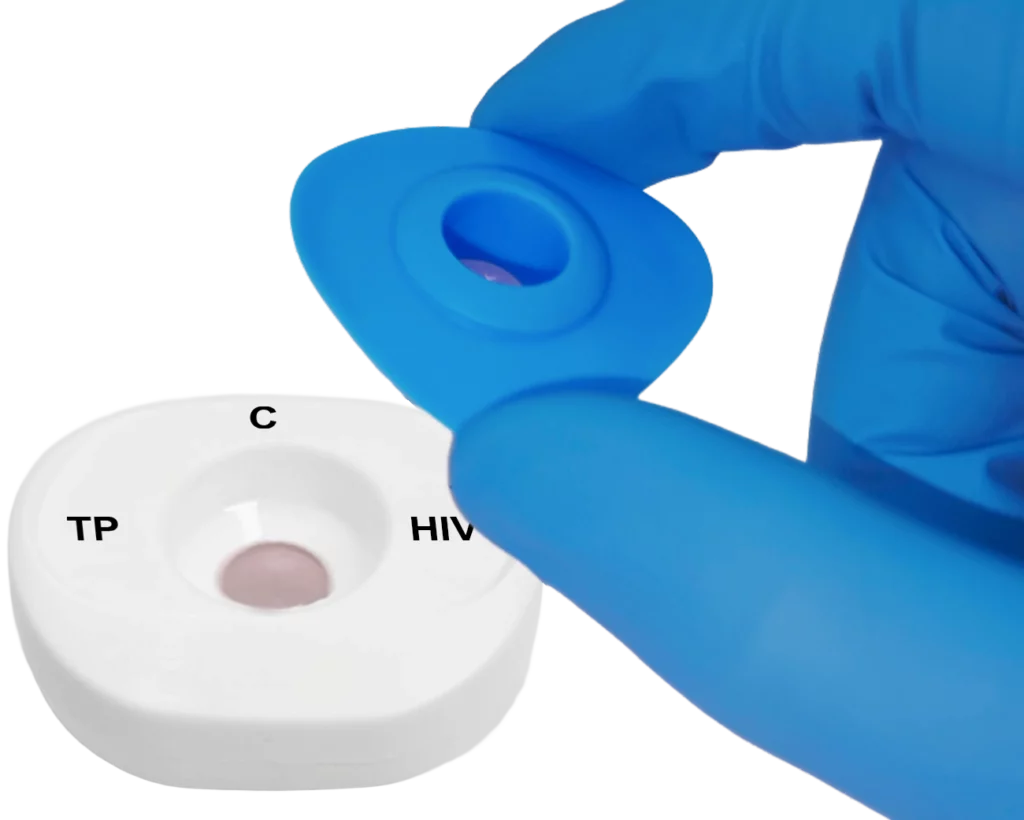
Place the InstantGold™ cap on the cartridge.
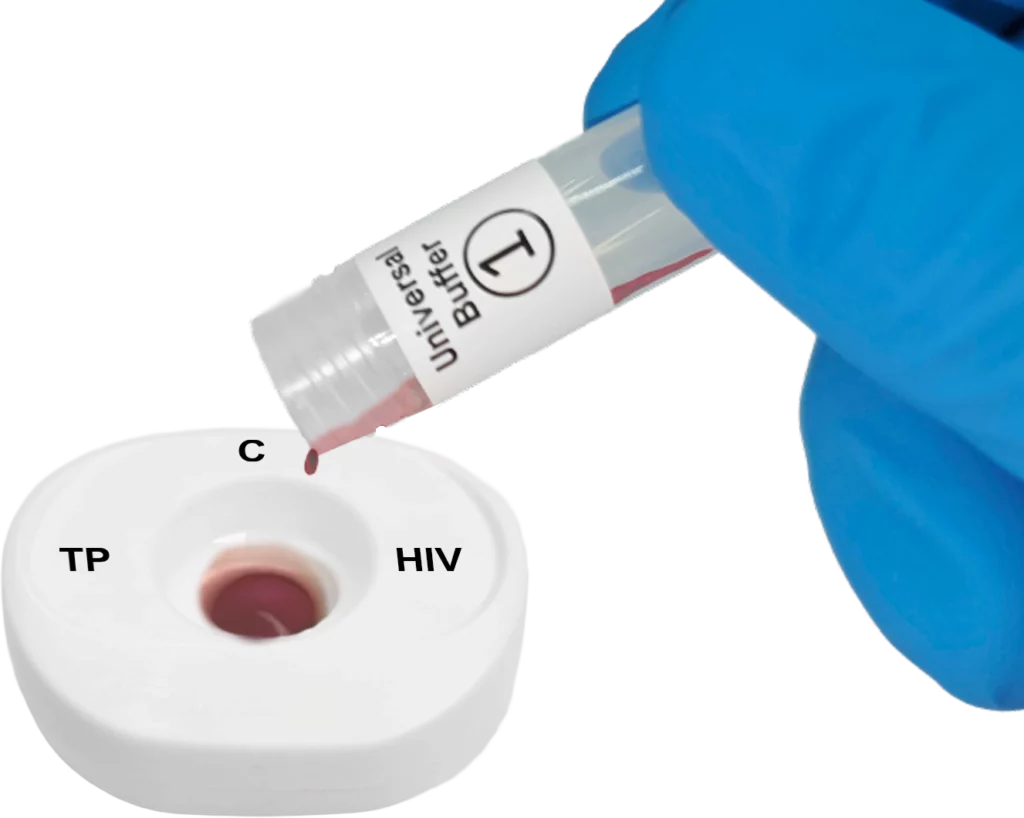
Pour lysed blood specimen into the test cartridge.
For a detailed walkthrough, watch video (Youtube, 5:48 min)
Proven Results, Trusted Performance
The Multiplo® Rapid TP/HIV Test consistently delivers high overall clinical performance in rigorous evaluations and inspections.
Sensitivity (PPA; overall)
Our test tailored to your needs
The Multiplo® Rapid TP/HIV Test is trusted in Canada for point-of-care use. Through regional distribution partners, we provide broad access to the POC format. It performs reliably in diverse and resource-limited settings without the need for specialized instruments, trained operators, or cold-chain storage. Our dual rapid test supports prenatal screening, public health STI programs, occupational exposure follow-up, counseling services, and mobile clinics – enabling earlier detection and reducing the risk of mother-to-child transmission.
This format includes a test tray in each pouch and is ideally suited to testing at the point of patient care, settings such as mobile clinics requiring a portable and all-in-one testing solution.
- Cat. No. 815311004902
- Qty. 20 tests per box
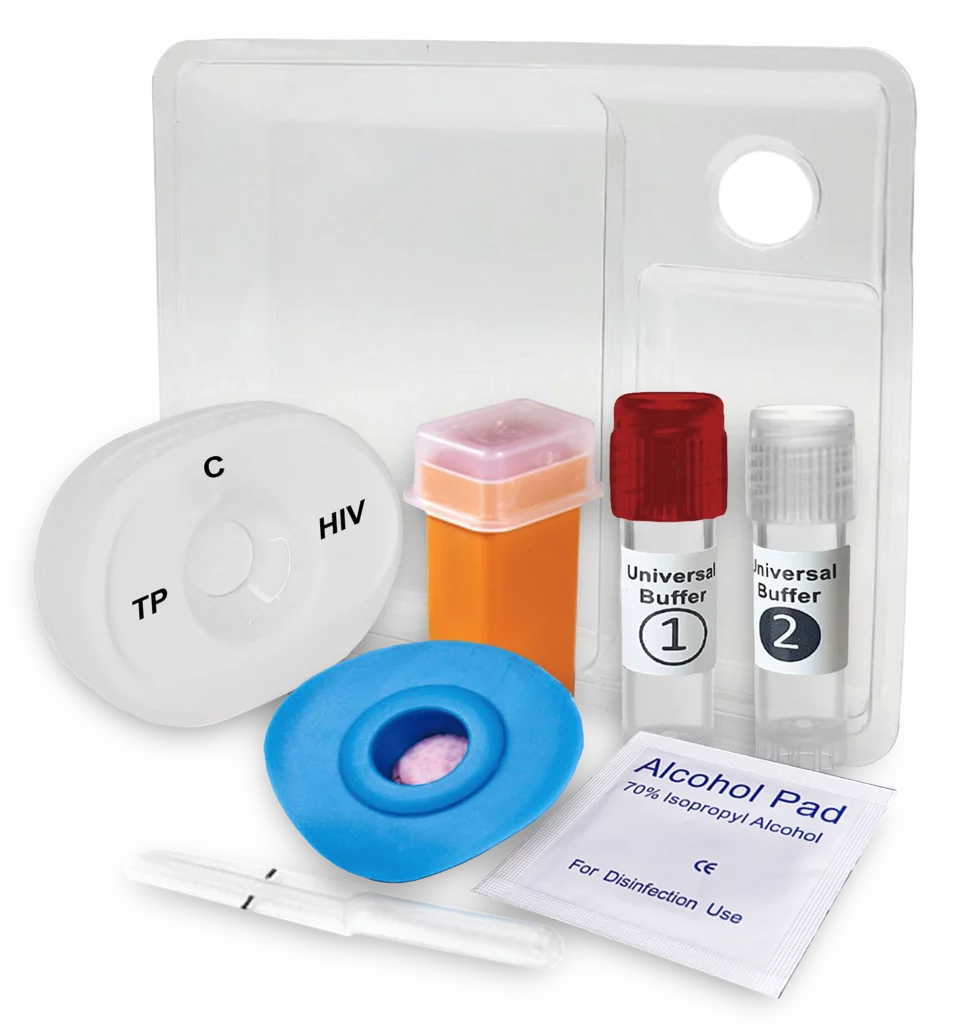
Downloads and Links
Additional materials are available through your partner login or upon request.
Product Specifications
Marketing Materials
Clinical Studies & Approvals
Further studies available upon request.
Related Products
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Miriad is intended for laboratory, academic, and research use only, and is not for use in diagnostic procedures.
Learn more about our upcoming technology and products advancing through our development pipeline.
Frequently Asked Questions
To learn more about Multiplo® Rapid TP/HIV Test, please contact us.
The test demonstrates 83.8% overall sensitivity for Syphilis (TP) antibodies, with a specificity of 99.2%. For higher-titer Syphilis samples (≥1:8), sensitivity is 97.3%, and for lower-titer samples (<1:8), sensitivity is 88.1%. For HIV-1/2, the test provides 100% sensitivity and 99.6% specificity.
Partners Contact Us
HIV and syphilis are closely linked, as they spread through similar routes and often share the same risk factors. Syphilis can also increase the risk of HIV transmission, making co-infections more likely. With rising infection rates reported by ECDC and WHO, early dual screening has become increasingly important. A combined test delivers fast, efficient and accurate results and is especially valuable in pregnancy, helping prevent complications and mother-to-child transmission. Dual rapid tests are also simpler, more cost-effective, and more comfortable for patients, requiring only a single blood sample.
Multiplo® Rapid TP/HIV Test uses MedMira’s unique Rapid Vertical Flow (RVF) technology™, which delivers instant results – significantly faster than conventional lateral flow assays. The RVF™ format requires only one buffer and features a single reaction window, reducing user steps and minimizing error potential. Unlike traditional tests that depend on strict timing or multiple wash steps, RVF™ provides timeless, easy-to-read results with no refrigeration or specialized equipment required. The technology offers laboratory-grade accuracy, low cost per test, and supports multiplexing for future assay expansion – combining speed, simplicity, and performance in a single platform.
RVF (Rapid Vertical Flow) is a flow-through technology that captures specific biomarkers from blood, serum, or plasma samples on a specialized membrane, delivering instant visual results.
Current Product Information for the U.S.
The Multiplo® Rapid TP/HIV Test is currently not authorized for diagnostic use under FDA regulations and is limited to non-diagnostic applications in the United States.
For customers in the U.S. seeking products for research use, we recommend our Miriad® Research Use Only (RUO) product line. Please refer to the Miriad® product pages for further details.
In addition to the Miriad® test portfolio, a single FDA-approved HIV-1/2 test is available for diagnostic use in the U.S. Please refer to the product page for further details and availability.

Explore your options with us
If you have any questions regarding the Multiplo® Rapid TP/HIV Test, its regulatory status in the U.S., or suitable research alternatives, please contact us for further information.
Current Product Information for the EU
The Multiplo® Rapid TP/HIV Test is currently not authorized for diagnostic use in the EU and is limited to non-diagnostic applications in CE-regulated markets.
For customers in the EU who require products for research applications, we recommend our Miriad® Research Use Only (RUO) product line. Please refer to the Miriad® RUO product pages for further details, and specifications.

Explore your options with us
If you have any questions regarding the Multiplo® Rapid TP/HIV Test, its regulatory status in the EU, or suitable research alternatives, please contact us for further information.
Outside North America, and the EU
one test - two results
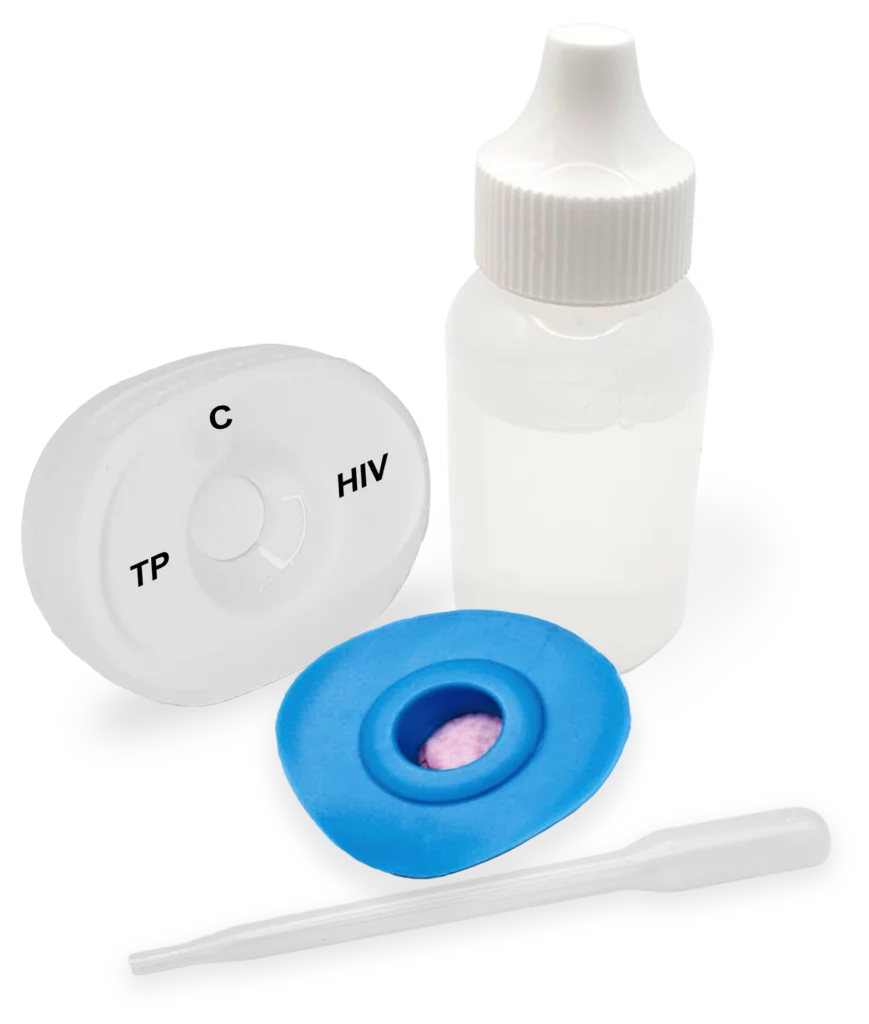
The Multiplo® Rapid TP/HIV Test enables simultaneous detection of antibodies to Treponema pallidum (syphilis) and HIV-1/2 in a single test using MedMira’s Rapid Vertical Flow (RVF) Technology™. Increasing co-infections – driven by shared risk factors and transmission routes – necessitate combined screening to support effective patient management and reduce mother-to-child transmission. It provides instant results in a straightforward three-step process and is available in different formats suitable for laboratory and point-of-care use. Its proven performance supports reliable testing and timely clinical and public health decision-making.
-
Immediate results
No waiting time, results do not vanish.
-
Reliable accuracy
Achieves high testing performance across multiple specimen matrices.
-
Multiplex testing
Multiple diseases tested on a single cartridge using one blood sample.
-
No equipment or refrigeration required
Stores at room temperature (2–30 °C / 35–86 °F) with a 12-month shelf life.
-
Easy to use and interpret
Simple procedure with instant, easy-to-read results, no special training required.
-
Ideal for any setting
Works with serum, plasma, and whole blood (venipuncture or fingerstick).

in three simple Steps
Testing doesn’t have to be complicated. Our intuitive three-step process ensures instant, consistent results with minimal effort.

Pour buffer onto the InstantGold™ cap.

Place the InstantGold™ cap on the cartridge.

Pour lysed blood specimen into the test cartridge.
For a detailed walkthrough, watch video (Youtube, 5:49 min)
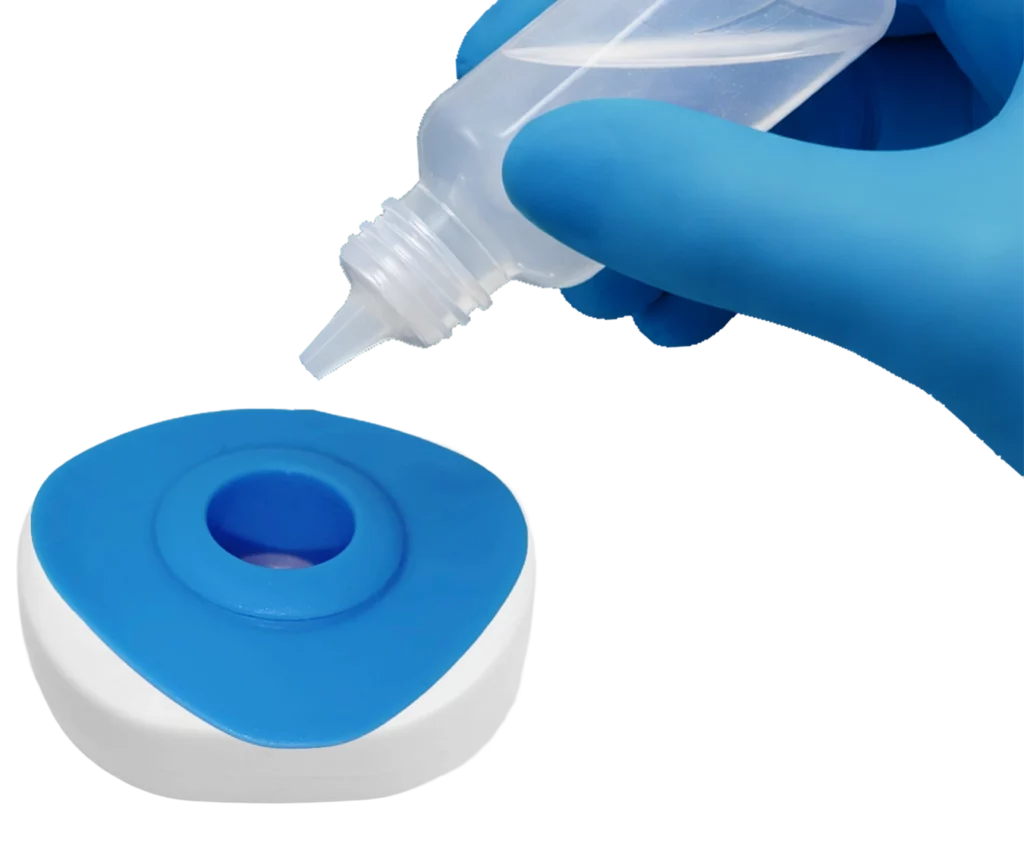
Place InstantGold™ cap on the test & add buffer.
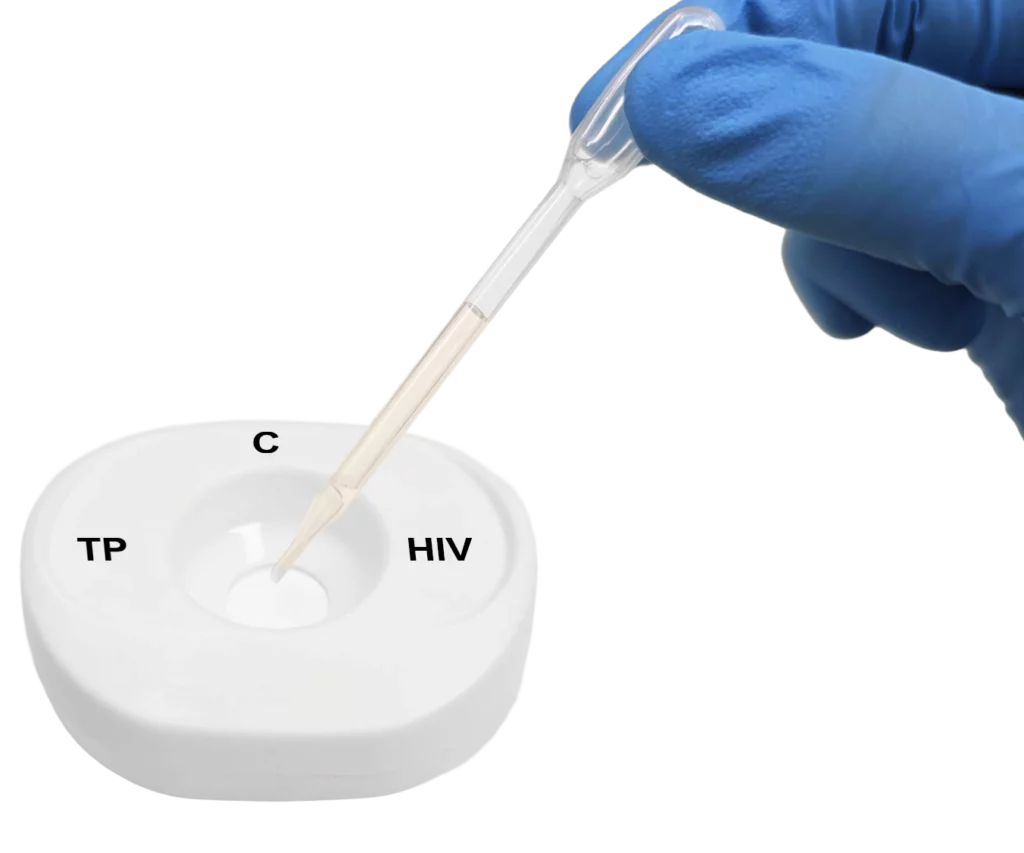
Apply specimen to the centre of the test membrane.
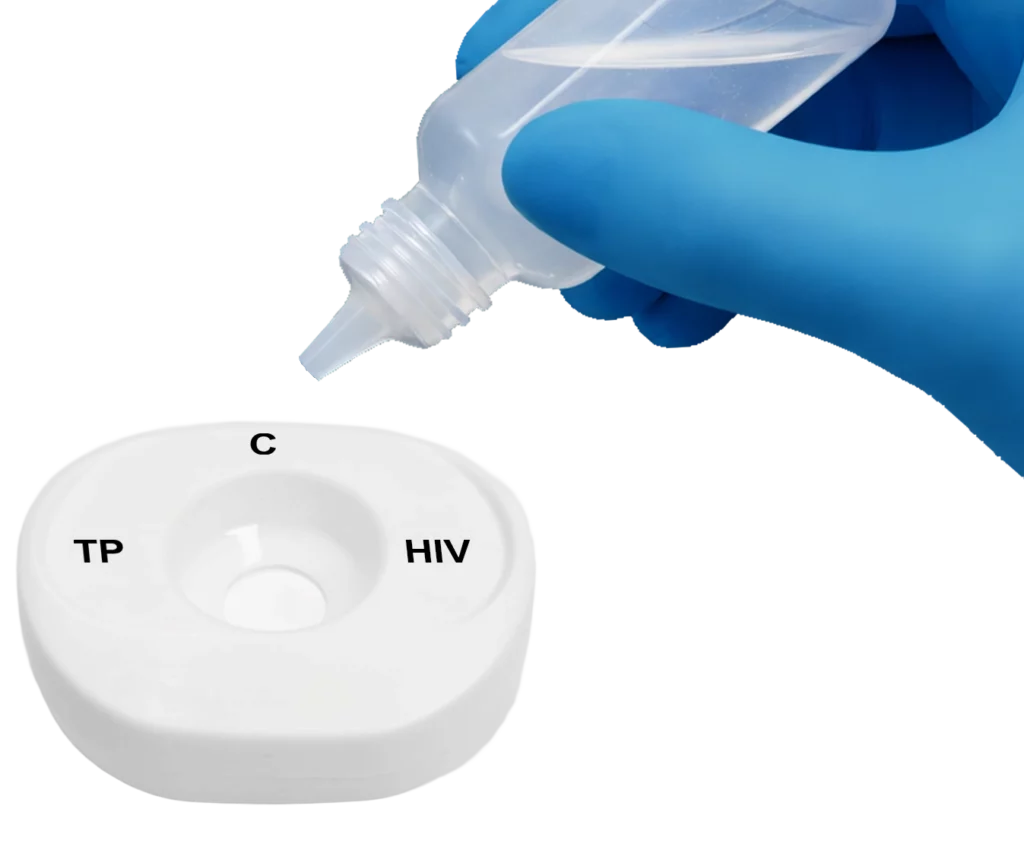
Apply buffer to the test center.
For a detailed walkthrough, watch video (Youtube, 2:11 min)
Proven Results, Trusted Performance
The Multiplo® Rapid TP/HIV Test consistently delivers high overall clinical performance in rigorous evaluations and inspections.
Sensitivity (PPA; overall)
Sensitivity (PPA; ≥1:8)
Our test tailored to your needs
The Multiplo® Rapid TP/HIV Test is trusted worldwide for both point-of-care and laboratory settings. Working with regional distribution partners, we provide broad access to three formats designed for different testing environments.
This format includes a test tray in each pouch and is ideally suited to testing at the point of patient care, settings such as mobile clinics requiring a portable and all-in-one testing solution.
- Qty. 20 tests per box
The LAB+ format is designed for laboratory use, offering maximum flexibility for testing serum, plasma, and venipuncture whole blood with reliable and consistent results.
- Qty. 50 tests per box
The LAB S/P format is designed for laboratory use, ideally suited for serum and plasma specimens and batch testing, ensuring efficient processing and dependable performance.
- Qty. 50 tests per box

Related Products
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Miriad is intended for laboratory, academic, and research use only, and is not for use in diagnostic procedures.
Learn more about our upcoming technology and products advancing through our development pipeline.
Frequently Asked Questions
To learn more about Multiplo® Rapid TP/HIV Test, please contact us.
The test demonstrates 83.8% overall sensitivity for Syphilis (TP) antibodies, with a specificity of 99.2%. For higher-titer Syphilis samples (≥1:8), sensitivity is 97.3%, and for lower-titer samples (<1:8), sensitivity is 88.1%. For HIV-1/2, the test provides 100% sensitivity and 99.6% specificity.
Partners Contact Us
HIV and syphilis are closely linked, as they spread through similar routes and often share the same risk factors. Syphilis can also increase the risk of HIV transmission, making co-infections more likely. With rising infection rates reported by ECDC and WHO, early dual screening has become increasingly important. A combined test delivers fast, efficient and accurate results and is especially valuable in pregnancy, helping prevent complications and mother-to-child transmission. Dual rapid tests are also simpler, more cost-effective, and more comfortable for patients, requiring only a single blood sample.
Multiplo® Rapid TP/HIV Test uses MedMira’s unique Rapid Vertical Flow (RVF) technology™, which delivers instant results – significantly faster than conventional lateral flow assays. The RVF™ format requires only one buffer and features a single reaction window, reducing user steps and minimizing error potential. Unlike traditional tests that depend on strict timing or multiple wash steps, RVF™ provides timeless, easy-to-read results with no refrigeration or specialized equipment required. The technology offers laboratory-grade accuracy, low cost per test, and supports multiplexing for future assay expansion – combining speed, simplicity, and performance in a single platform.
RVF™ (Rapid Vertical Flow™) is a flow-through technology that captures specific biomarkers from blood, serum, or plasma samples on a specialized membrane, delivering instant visual results.



















