rapid HCV/HIV antibody test
Accurate, rapid simultaneous detection of antibodies to HIV-1/2 and/or Hepatitis C virus (HCV) in whole blood, serum, and plasma specimens.
one test - two results
The Multiplo® Rapid HCV/HIV Antibody Test enables simultaneous detection of Human Immunodeficiency Virus Type 1 and Type 2 (anti-HIV-1/2), and antibodies to Hepatitis C virus (anti-HCV), using MedMira’s Rapid Vertical Flow (RVF) Technology™. Its simple and proven three-step procedure delivers fast, reliable results.
-
Immediate results
No waiting time, results do not vanish.
-
Reliable accuracy
Achieves high HCV testing performance across multiple specimen matrices.
-
Multiplex testing
Multiple diseases tested on a single cartridge using one blood sample.
-
No equipment or refrigeration required
Stores at room temperature (2–30 °C / 35–86 °F) with a 12-month shelf life.
-
Easy to use and interpret
Simple procedure with instant, easy-to-read results, no special training required.
-
Ideal for any setting
Works with serum, plasma, and whole blood (venipuncture or fingerstick).

in three simple Steps
Testing doesn’t have to be complicated. Our intuitive three-step process ensures instant, consistent results with minimal effort.
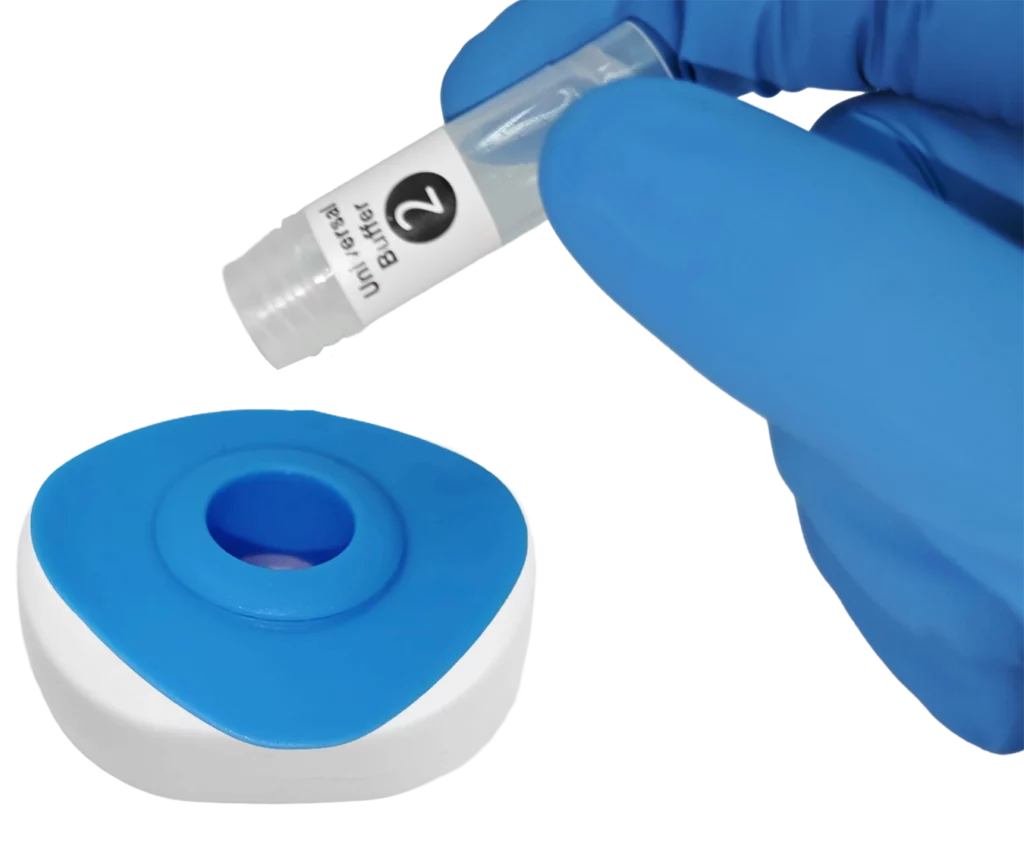
Pour buffer onto the InstantGold™ cap.
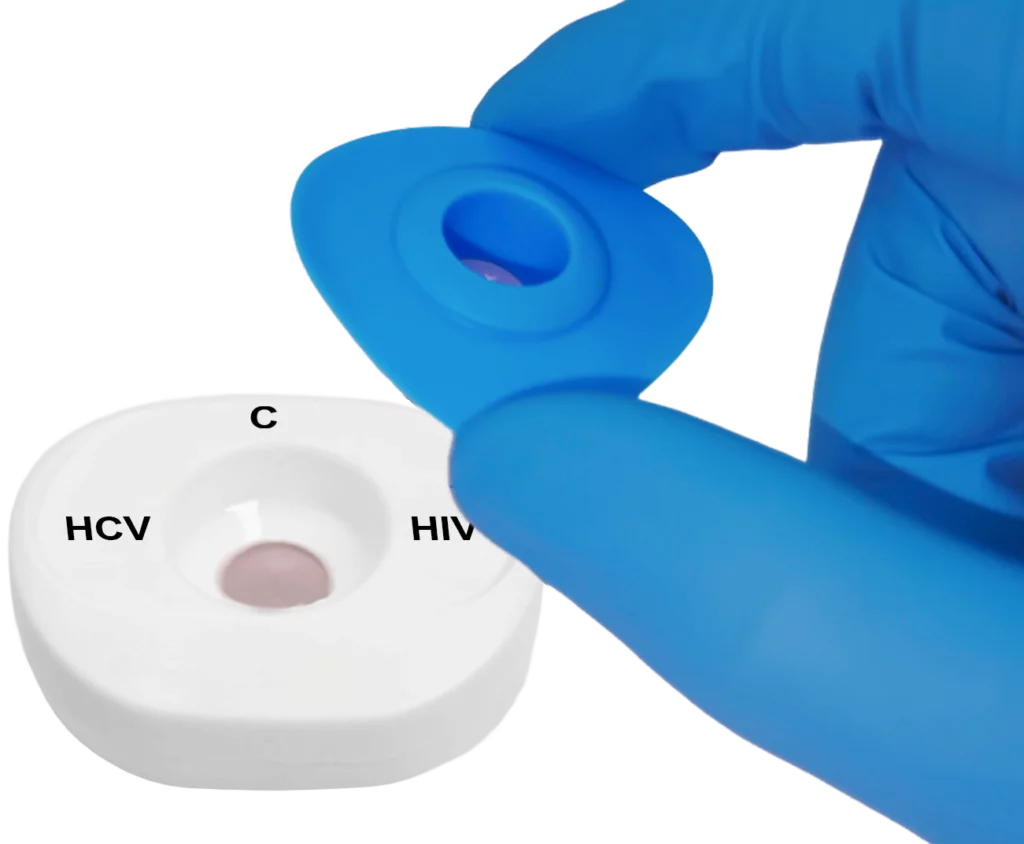
Place the InstantGold™ cap on the cartridge.
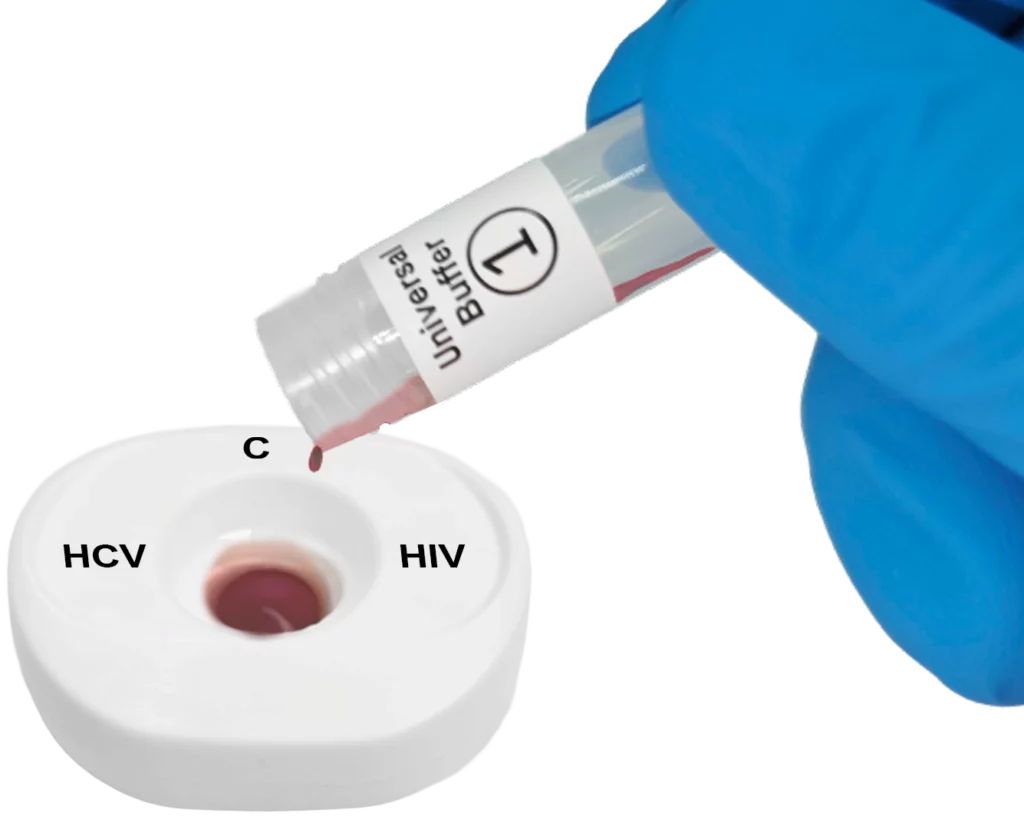
Pour lysed blood specimen into the test cartridge.
For a similar procedure, watch the Multiplo® Rapid TP/HIV Antibody Test video (Youtube, 5:49 min).
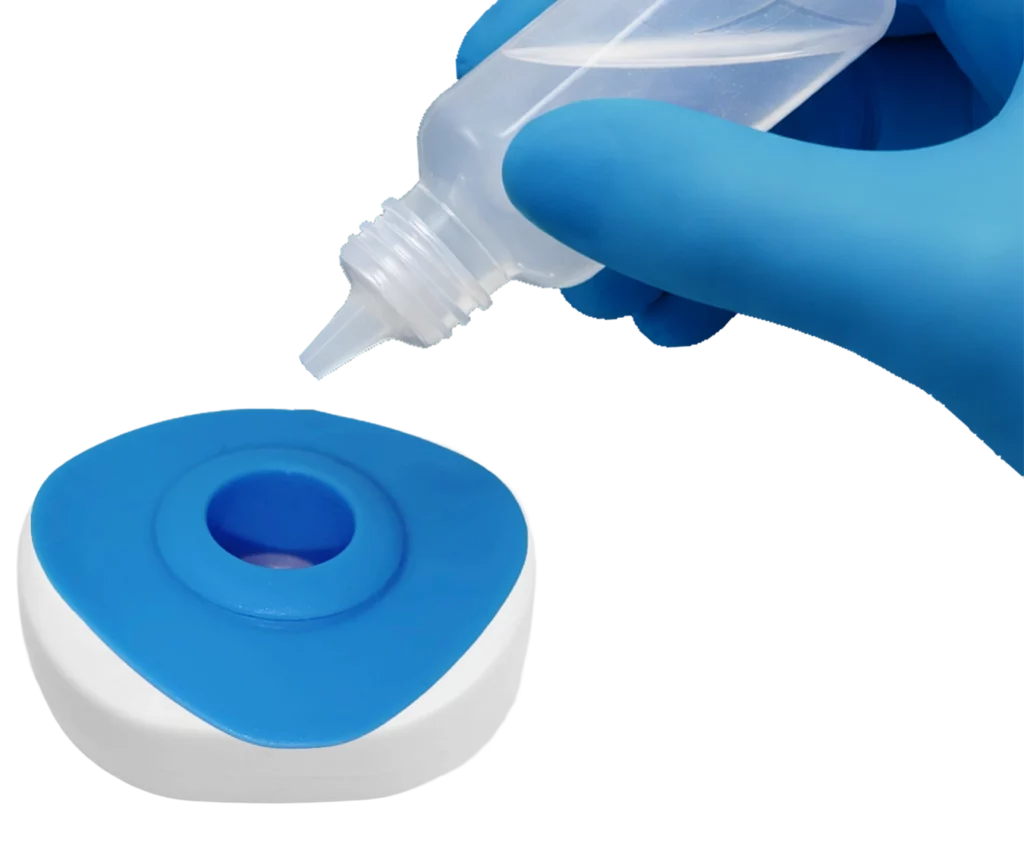
Place InstantGold™ cap on the test & add buffer.
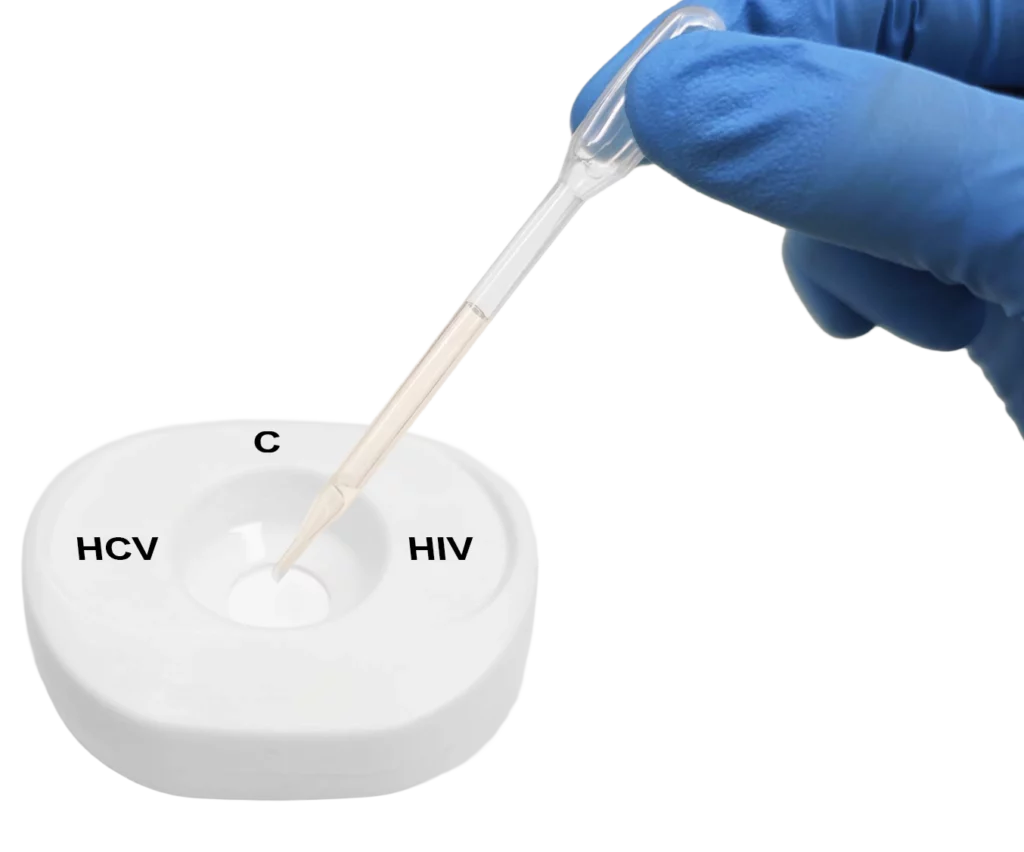
Apply specimen to the centre of the test membrane.
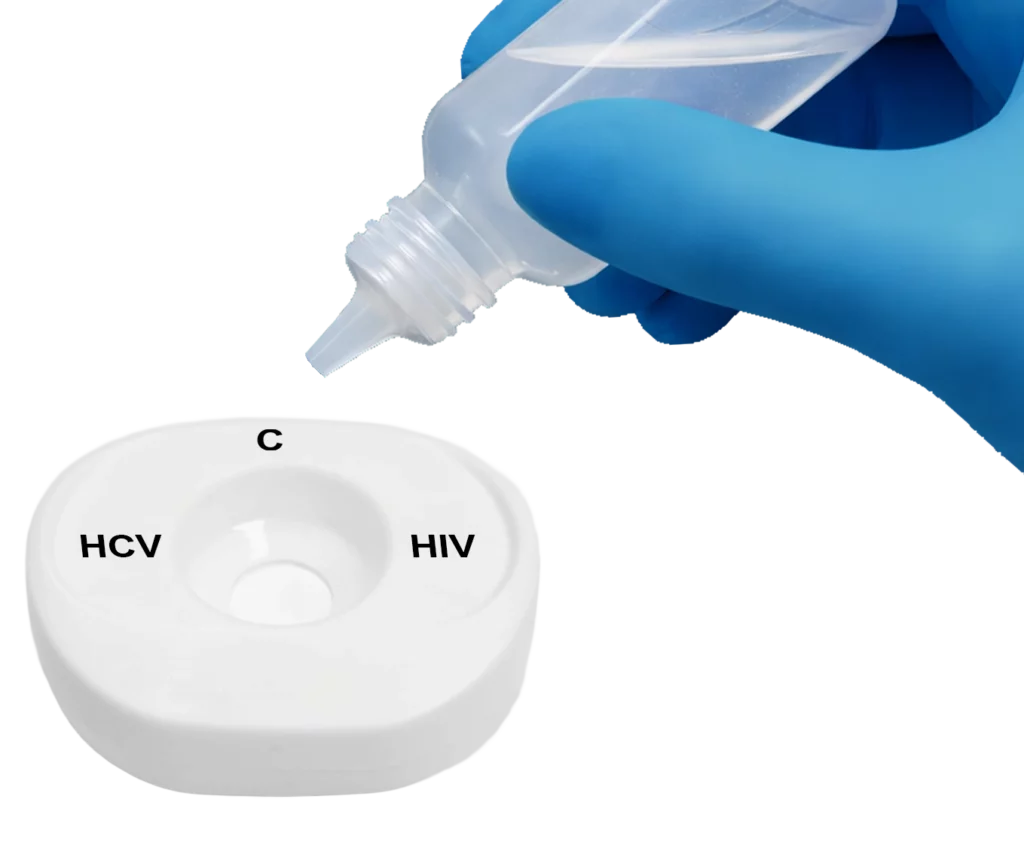
Apply buffer to the test center.
For a similar procedure, watch the Multiplo® Rapid TP/HIV Antibody Test video (Youtube, 2:11 min).
Explore your options with us
This product is available for international applications outside North America and the EU. For information regarding Multiplo® Rapid HCV/HIV Antibody Test, including availability, ordering procedures, and technical documentation, please contact our team.
Related Products
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Miriad is intended for laboratory, academic, and research use only, and is not for use in diagnostic procedures.
Learn more about our upcoming technology and products advancing through our development pipeline.
Frequently Asked Questions
To learn more about Multiplo® Rapid HCV/HIV Antibody Test, please contact us.
Learn more
Rapid tests provide valuable advantages across a range of environments, from point-of-care settings to research and laboratory workflows. They deliver results within minutes without relying on extensive infrastructure, specialized instruments, or highly trained personnel, which helps reduce costs and streamline testing processes. Modern rapid assays often demonstrate performance comparable to laboratory methods, making them a practical option for decentralized screening, high-throughput research studies, emergency situations, and programs that require immediate decision-making or broad accessibility.
Contact Us
HCV and HIV often share transmission routes, particularly in contexts involving unprotected sexual contact or exposure to infected blood. A combined test streamlines screening by eliminating the need for multiple assays, improving workflow efficiency in both laboratory and point-of-care settings. It simplifies sample handling while providing broad, reliable detection for these key infectious diseases. The Multiplo® Rapid HCV/HIV Antibody Test supports effective screening through a simple three-step procedure, delivering dependable results with minimal hands-on time.
Yes. Our Multiplo® product line offers testing for several diseases within a single test cartridge and includes assays such as the Multiplo® Rapid TP/HIV Test, which holds regulatory approvals in Canada (home country approval). In addition, our Miriad® product line – available for research use only – provides further testing options, including assays capable of detecting HCV and HIV.



















