Complete Syphilis (TP/nTP) antibody test
Combined detection of treponemal (TP) and non-treponemal (nTP) antibodies in one test.
Not yet Health Canada approved
Answers in real time
The Multiplo® Complete Syphilis (TP/nTP) Antibody Test detects both treponemal (TP) and non-treponemal (nTP) antibodies in one assay, combining screening and confirmation in a single device. The TP component of the test shows past or current exposure to the syphilis bacterium, while the nTP screening test reflects antibody levels that rise during an active infection. By identifying biomarkers of active and past infection, the test provides a comprehensive, accurate, and cost-efficient testing solution that meets global testing needs. It delivers instant results in a simple three-step process.
-
Immediate results
No waiting time, results do not vanish.
-
Reliable accuracy
Achieves high testing performance.
-
Comprehensive testing solution
Identifies biomarkers of active and past infection for a complete profile.
-
No equipment or refrigeration required
Stores at room temperature (2–30 °C / 35–86 °F) with a 12-month shelf life.
-
Easy to use and interpret
Simple procedure with instant, easy-to-read results, no special training required.
-
Ideal for POC setting
Works with whole blood (fingerstick).

in three simple Steps
Testing doesn’t have to be complicated. Our intuitive three-step process ensures instant, consistent results with minimal effort.
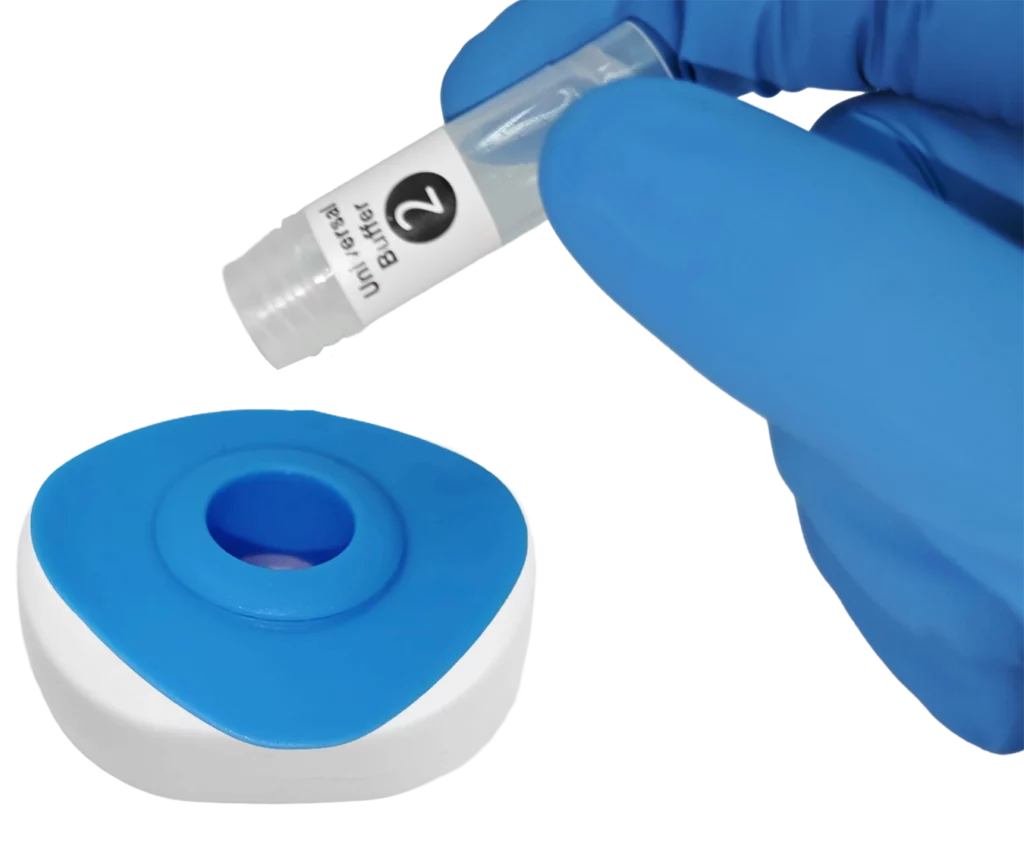
Pour buffer onto the InstantGold™ cap.

Place the InstantGold™ cap on the cartridge.
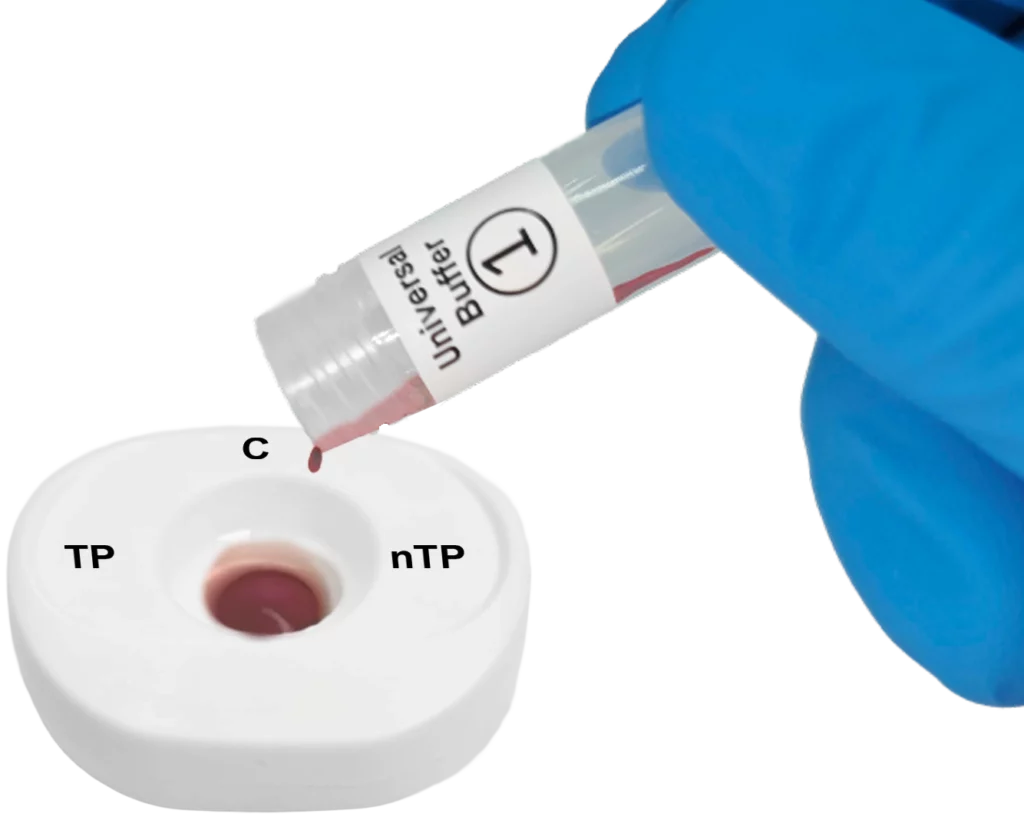
Pour lysed blood specimen into the test cartridge.
For a detailed walkthrough, watch video (Youtube, 4:20 min)
In a real-world STI clinic setting, the MedMira Multiplo® Complete Syphilis (TP/nTP) test demonstrated strong clinical performance and meaningful impact on patient management. Beyond enabling more rapid identification of infectious syphilis, the availability of a non-treponemal result at the point of care supported evidence-based decisions to withhold unnecessary antibiotic treatment. When used alongside clinical assessment and established staging algorithms, dual TP/nTP point-of-care testing can improve both timely treatment and antibiotic stewardship.
Patrick O’Byrne, NP PhD
Professor of Nursing, University of Ottawa
Nurse Practitioner, Ottawa Public Health Sexual Health Clinic
Related Products
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Miriad is intended for laboratory, academic, and research use only, and is not for use in diagnostic procedures.
Learn more about our upcoming technology and products advancing through our development pipeline.
Frequently Asked Questions
To learn more about Multiplo® Complete Syphilis (TP/nTP) Antibody Test, please contact us.
RVF™ (Rapid Vertical Flow™) is a flow-through technology that captures specific biomarkers from blood, serum, or plasma samples on a specialized membrane, delivering instant visual results.
Learn more
Combined treponemal (TP) and non-treponemal (nTP) testing provides a more complete and reliable assessment of syphilis infection. TP tests show whether someone has ever been exposed to Treponema pallidum, while nTP tests indicate whether the infection is currently active. Using both markers in a single test improves diagnostic accuracy, helps distinguish active from past or treated infections, and supports faster, more informed clinical decisions. This also helps avoid unnecessary treatment of patients who are TP-positive but do not have an active infection, preventing inappropriate use of antibiotics.
You can purchase the Multiplo® Complete Syphilis (TP/nTP) Antibody Test through authorized distributors. For purchasing information outside North America and the EU, please contact the sales team at sales@medmira.com.
Please note that the test is currently in clinical trials and has not yet been approved by Health Canada. In Canada, it is available for research use only and is not intended for diagnostic purposes. Commercial availability in Canada will begin once the necessary regulatory approvals have been granted.
Multiplo® Complete Syphilis (TP/nTP) Antibody Test uses MedMira’s unique Rapid Vertical Flow (RVF) technology™, which delivers instant results – significantly faster than conventional lateral flow assays. The RVF™ format requires two buffers and features a single reaction window, reducing user steps and minimizing error potential. Unlike traditional tests that depend on strict timing or multiple wash steps, RVF™ provides timeless, easy-to-read results with no refrigeration or specialized equipment required. The technology offers laboratory-grade accuracy, low cost per test, and supports multiplexing for future assay expansion – combining speed, simplicity, and performance in a single platform.
Rapid tests provide valuable advantages across a range of environments, from point-of-care settings to research and laboratory workflows. They deliver results within minutes without relying on extensive infrastructure, specialized instruments, or highly trained personnel, which helps reduce costs and streamline testing processes. Modern rapid assays often demonstrate performance comparable to laboratory methods, making them a practical option for decentralized screening, high-throughput research studies, emergency situations, and programs that require immediate decision-making or broad accessibility.
Current Product Information for the U.S.
The Multiplo® Complete Syphilis (TP/nTP) Antibody Test is currently not authorized for diagnostic use under FDA regulations and is limited to non-diagnostic applications in the United States.
For customers in the U.S. seeking products for research use, we recommend our Miriad® Research Use Only (RUO) product line. Please refer to the Miriad® product pages for further details.
In addition to the Miriad® test portfolio, a single FDA-approved HIV-1/2 test is available for diagnostic use in the U.S. Please refer to the product page for further details and availability.

Explore your options with us
If you have any questions regarding the Multiplo® Complete Syphilis (TP/nTP) Antibody Test, its regulatory status in the U.S., or suitable research alternatives, please contact us for further information.
Current Product Information for the EU
The Multiplo® Complete Syphilis (TP/nTP) Antibody Test is currently not authorized for diagnostic use in the EU and is limited to non-diagnostic applications in CE-regulated markets.
For customers in the EU who require products for research applications, we recommend our Miriad® Research Use Only (RUO) product line. Please refer to the Miriad® RUO product pages for further details, and specifications.

Explore your options with us
If you have any questions regarding the Multiplo® Complete Syphilis (TP/nTP) Antibody Test, its regulatory status in the EU, or suitable research alternatives, please contact us for further information.
Outside North America, and the EU
Answers in real time
The Multiplo® Complete Syphilis (TP/nTP) Antibody Test detects both treponemal (TP) and non-treponemal (nTP) antibodies in one assay, combining screening and confirmation in a single device. The TP component of the test shows past or current exposure to the syphilis bacterium, while the nTP screening test reflects antibody levels that rise during an active infection. By identifying biomarkers of active and past infection, the test provides a comprehensive, accurate, and cost-efficient testing solution that meets global testing needs. It delivers instant results in a simple three-step process and is available in point-of-care format for use by Healthcare Professionals. Its proven performance supports reliable testing and timely clinical and public health decision-making.
-
Immediate results
No waiting time, results do not vanish.
-
Reliable accuracy
Achieves high testing performance for fingerstick whole blood.
-
Comprehensive testing solution
Identifies biomarkers of active and past infection for a complete profile.
-
No equipment or refrigeration required
Stores at room temperature (2–30 °C / 35–86 °F) with a 12-month shelf life.
-
Easy to use and interpret
Simple procedure with instant, easy-to-read results, no special training required.
-
Ideal for POC setting
Works with whole blood (fingerstick).

in three simple Steps
Testing doesn’t have to be complicated. Our intuitive three-step process ensures instant, consistent results with minimal effort.

Pour buffer onto the InstantGold™ cap.

Place the InstantGold™ cap on the cartridge.

Pour lysed blood specimen into the test cartridge.
For a detailed walkthrough, watch video (Youtube, 4:20 min)
In a real-world STI clinic setting, the MedMira Multiplo® Complete Syphilis (TP/nTP) test demonstrated strong clinical performance and meaningful impact on patient management. Beyond enabling more rapid identification of infectious syphilis, the availability of a non-treponemal result at the point of care supported evidence-based decisions to withhold unnecessary antibiotic treatment. When used alongside clinical assessment and established staging algorithms, dual TP/nTP point-of-care testing can improve both timely treatment and antibiotic stewardship.
Patrick O’Byrne, NP PhD
Professor of Nursing, University of Ottawa
Nurse Practitioner, Ottawa Public Health Sexual Health Clinic
Our test tailored to your needs
The Multiplo® Complete Syphilis (TP/nTP) Antibody Test is a trusted point-of-care solution worldwide, delivered with broad accessibility through our network of regional distribution partners.
This format includes a test tray in each pouch and is ideally suited to testing at the point of patient care, settings such as mobile clinics requiring a portable and all-in-one testing solution.
- Qty. 20 tests per box
Related Products
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Designed for use across POC settings, laboratories, academic institutions, and research environments.
Miriad is intended for laboratory, academic, and research use only, and is not for use in diagnostic procedures.
Learn more about our upcoming technology and products advancing through our development pipeline.
Frequently Asked Questions
To learn more about Multiplo® Complete Syphilis (TP/nTP) Antibody Test, please contact us.
RVF™ (Rapid Vertical Flow™) is a flow-through technology that captures specific biomarkers from blood, serum, or plasma samples on a specialized membrane, delivering instant visual results.
Learn more
Not at this time. Multiplo® Complete Syphilis (TP/nTP) Antibody Test availability in the United States will begin once the appropriate regulatory clearance has been granted. The Miriad® Complete Syphilis (TP/nTP) Antibody Test can be purchased in the United States; however, it is available for research use only and not for diagnostic purposes.
You can purchase the Multiplo® Complete Syphilis (TP/nTP) Antibody Test through authorized distributors. For purchasing information outside North America and the EU, please contact the sales team at sales@medmira.com.
Please note that the test is currently in clinical trials and has not yet been approved by Health Canada. In Canada, it is available for research use only and is not intended for diagnostic purposes. Commercial availability in Canada will begin once the necessary regulatory approvals have been granted.
Rapid tests provide valuable advantages across a range of environments, from point-of-care settings to research and laboratory workflows. They deliver results within minutes without relying on extensive infrastructure, specialized instruments, or highly trained personnel, which helps reduce costs and streamline testing processes. Modern rapid assays often demonstrate performance comparable to laboratory methods, making them a practical option for decentralized screening, high-throughput research studies, emergency situations, and programs that require immediate decision-making or broad accessibility.
Multiplo® Complete Syphilis (TP/nTP) Antibody Test uses MedMira’s unique Rapid Vertical Flow (RVF) technology™, which delivers instant results – significantly faster than conventional lateral flow assays. The RVF™ format requires two buffers and features a single reaction window, reducing user steps and minimizing error potential. Unlike traditional tests that depend on strict timing or multiple wash steps, RVF™ provides timeless, easy-to-read results with no refrigeration or specialized equipment required. The technology offers laboratory-grade accuracy, low cost per test, and supports multiplexing for future assay expansion – combining speed, simplicity, and performance in a single platform.



















