MedMira receives investigational testing authorization for its Multiplo® Complete Syphilis (TP/nTP) Antibody Test
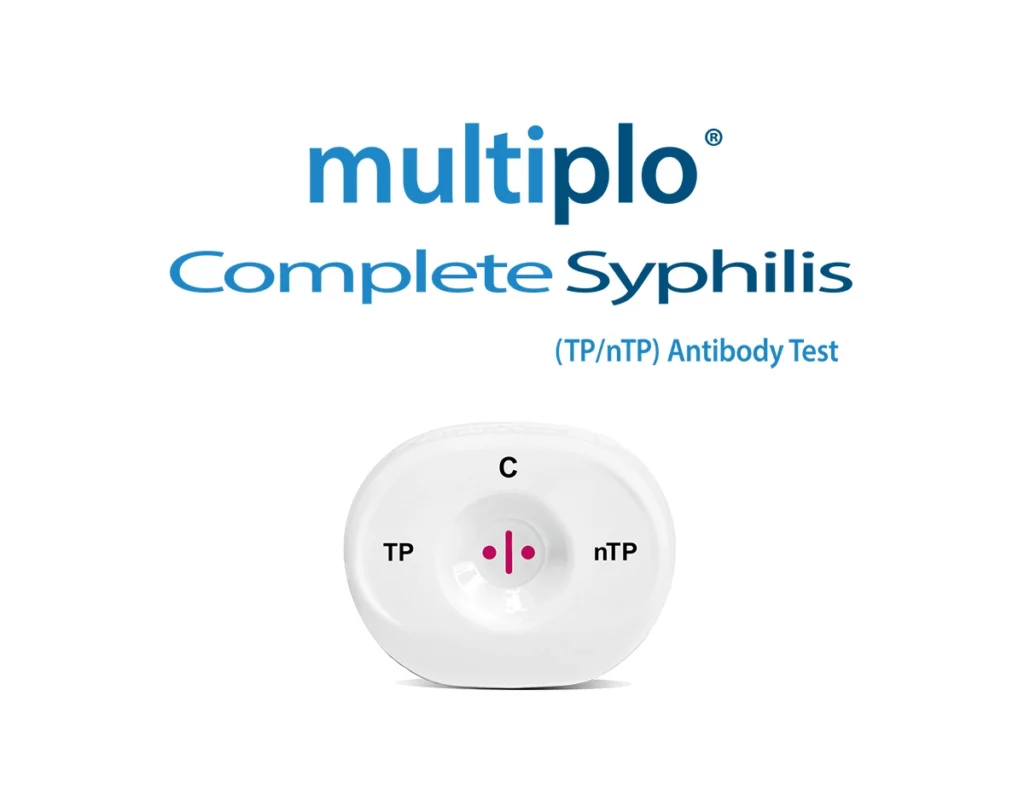
Halifax, Nova Scotia, 9 January, 2025 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announced that its received investigational testing authorization (ITA) from Health Canada to begin clinical trials for its Multiplo® Complete Syphilis (TP/nTP) Antibody Test (Multiplo® TP/nTP), a cutting edge device that can determine active and non-active syphilis. The upcoming trials will be led […]
Clinical Trials to get Underway with Multiplo® Complete Syphilis (TP/nTP) Antibody Test – A Potential Game Changer to Address Syphilis Health Crisis in Canada

Halifax, Nova Scotia, 13 August, 2024 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announces it will conduct full clinical trials for its unique Multiplo® Complete Syphilis (TP/nTP) Antibody Test (Multiplo® TP/nTP) with the goal of getting it licensed in Canada. The new Multiplo® TP/nTP will provide a complete system which combines screening and confirmation in […]
Test yourself in a smart way

A fact sheet for the importance of testing for both TP/nTP. Multiplo® Compete Syphilis (TP/nTP) Antibody Test – testing was never so easy and fast. Take YOUR health in YOUR hands. Download TP/nTP general public fact sheet
MedMira receives CE mark for Multiplo® Complete Syphilis (TP/nTP) Antibody Test

Halifax, Nova Scotia, 12 May, 2022 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announces the achievement of another milestone in its regulatory strategy by receiving the CE mark for its Multiplo® Complete Syphilis (TP/nTP) Antibody Test (Multiplo® TP/nTP). This approval allows the Company to further strengthen its product offering in all markets accepting the CE […]



















