MedMira receives 3 new trademarks

MedMira has been notified that the Company has successfully defended its claim on its submitted trademarks. VYRAU.S. Serial Number: 90407022 BYRAU.S. Serial Number: 90407042 PYRAU.S. Serial Number: 90407030 Registration certification will be issued in the coming weeks.
Finger-Prick Test Developed For ‘Trich’ A Common, Undiagnosed STI
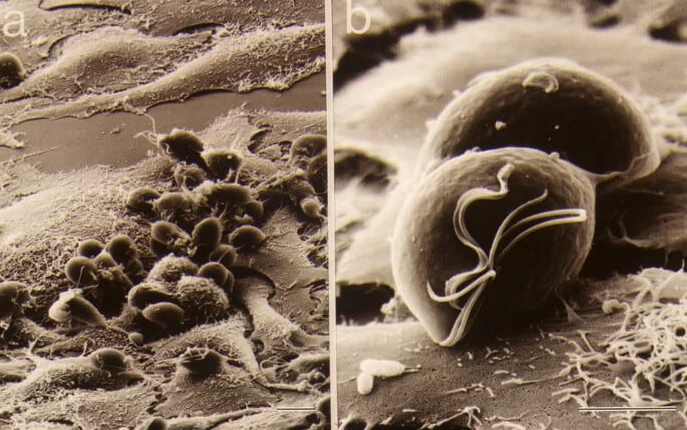
Washington State University: Link to full article (WSU News) A quick, affordable diagnostic test developed by a Washington State University researcher may help curb one of the most prevalent but least discussed sexually transmitted infections. More common than chlamydia or gonorrhea, Trichomonas vaginalis, also known as trich, causes no symptoms in about 70% of those […]
MedMira receives CE mark for its VYRA™ CoV2Flu Antigen Test

Halifax, Nova Scotia, 11 January, 2023 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announces the receipt of the CE mark for its VYRA™ CoV2Flu Antigen Test (VYRA™ CoV2Flu), the fastest combination antigen test available to detect SARS-CoV-2, Flu A and Flu B. The Company received the CE mark and VYRA™ CoV2Flu is now available in […]
MedMira’s G4 HIV (FDA approved) rapid test – an independent evaluation by the CDC

Performance Evaluation of the MedMira Reveal G4 LAB S/P and POC HIV Antibody Rapid Screening Tests using Plasma and Whole Blood Specimens Rebecca Rossetti1, Tara Smith2, Wei Luo1, Silvina Masciotra1 1Division of HIV/AIDS Prevention, Centers for Disease Control and Prevention2Oak Ridge Institute for Science and Education Download the full study
Point-of-Care Diagnostic for Trichomonas vaginalis based on MedMira’s RVF Technology™
Halifax, Nova Scotia, 3 January, 2023 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announces the publication of Point-of-Care Diagnostic for Trichomonas vaginalis, the Most Prevalent, Non-Viral Sexually Transmitted Infection – (available: https://www.mdpi.com/2076-0817/12/1/77), in the special edition of Pathogens by MDPI, a Swiss based medical journal. The article written by Professor John Alderete from the School […]
MedMira Introduces VYRA TriDemic

Halifax, Nova Scotia, 29 December, 2022 – Today, MedMira Inc. (MedMira) (TSXV: MIR) presents its latest addition to the VYRA™ product line, the VYRA™ TriDemic Antigen Rapid Test for the simultaneous detection of SARS-CoV-2, Flu A, Flu B and Respiratory Syncytial Virus (RSV). VYRA™ TriDemic, a four-in-one test, which is a direct response to the […]
MedMira’s Multiplo TP/HIV Rapid Test: One Test, More Answers
A study completed by the Indian Red Cross Blood Society showed that MedMira’s Multiplo® TP/HIV Rapid Test is your immediate quality answer. MedMira’s Multiplo® TP/HIV Rapid Test. One Test, More Answers. Human immunodeficiency virus (HIV) is a blood-borne virus that poses a significant threat to global health, especially given that current therapies for HIV infection […]
Dr. Hermes Chan, CEO, Co-Founder and Co-Inventor receives Queen’s Jubilee Platinum Medal

Rafah DiCostanzo, MLA for Clayton Park West acknowledges outstanding constituents and has proposed Dr. Hermes Chan, CEO, Co-Founder and Co-Inventor to receive the Queen’s Jubilee Platinum Medal for his essential work during the pandemic. MedMira Inc. had developed four rapid tests specific for SARS-CoV-2 based on its patented Rapid Vertical Flow Technology during the pandemic, […]
MedMira receives CE mark for its VYRA™ COVID-19 Antigen Test

Halifax, Nova Scotia, 14 October, 2022 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announces the receipt of the CE mark for its VYRA™ COVID-19 Antigen Test (VYRA™ COVID-19), the fastest SARS-CoV-2 Antigen test available. The Company received the CE mark today and is now available in Europe and any countries accepting the CE mark. MedMira […]
Test yourself in a smart way
A fact sheet for the importance of testing for both TP/nTP. Multiplo® Compete Syphilis (TP/nTP) Antibody Test – testing was never so easy and fast. Take YOUR health in YOUR hands. Download TP/nTP general public fact sheet



















