Finger-Prick Test Developed For ‘Trich’ A Common, Undiagnosed STI
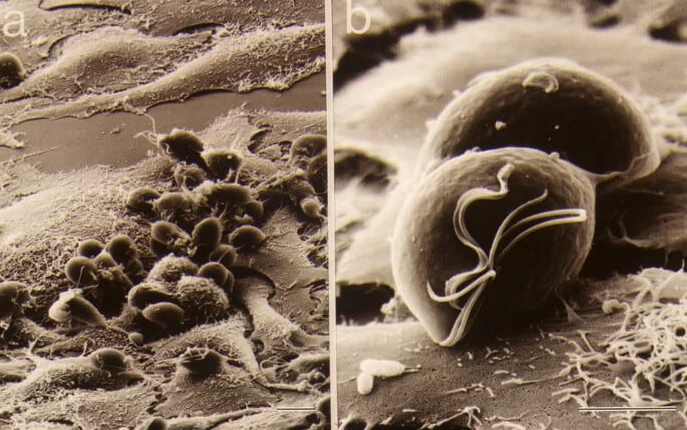
Washington State University: Link to full article (WSU News) A quick, affordable diagnostic test developed by a Washington State University researcher may help curb one of the most prevalent but least discussed sexually transmitted infections. More common than chlamydia or gonorrhea, Trichomonas vaginalis, also known as trich, causes no symptoms in about 70% of those […]
MedMira provides Progress Update

Halifax, Nova Scotia, 30 September, 2022 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announces the appointment of Dr. Sam Ratnam as MedMira’s Director of Scientific & Regulatory Affairs and provides an update on its regulatory progress in the U.S. market. U.S. Regulatory Update on COVID-19 Products The recent announced recommendation by the U.S. FDA to […]
Multiplo® Product Line – A Simple Solution in One

MedMira’s unique Multiplo® Product Line: Did you know, MedMira is working with a number of cruise lines which offer MedMira’s Multiplo® products on their ships for immediate screening of various diseases. The easy-to-use and fast handling is the perfect all-in-one test in ANY setting.
VYRA™ Product Line Update

Halifax, Nova Scotia, 20 May, 2022 – Today, MedMira Inc. (MedMira) (TSXV: MIR) provides an update on the Company’s regulatory work for Canada for its VYRA™ product line. On the 10th of May 2022, Health Canada issued to all stakeholder further information about the current Interim Order No. 3. As a result, the regulators have […]
MedMira receives CE mark for Multiplo® Complete Syphilis (TP/nTP) Antibody Test
Halifax, Nova Scotia, 12 May, 2022 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announces the achievement of another milestone in its regulatory strategy by receiving the CE mark for its Multiplo® Complete Syphilis (TP/nTP) Antibody Test (Multiplo® TP/nTP). This approval allows the Company to further strengthen its product offering in all markets accepting the CE […]
MedMira receives the CE mark for its Reveal® TP (Syphilis) and starts clinical trials in Canada

Halifax, Nova Scotia, 9 March, 2022 – Today, MedMira Inc. (MedMira) (TSXV: MIR) launches its Reveal® TP (Syphilis) product in Europe and gets clinical trial underway in Canada. The Company received the CE mark today and is now available in Europe and any countries accepting the CE mark. Furthermore, the Company continues their work on […]
Corporate update

In the second financial quarter of FY2022, MedMira continued its strategic plan to focus its own product portfolio on the two fastest growing disease segments in the IVD rapid test market. Furthermore, the Company continued its work with public and private partners to develop new potential tests based on its Rapid Vertical Flow (RVF) Technology™ […]
MedMira Provides a Pre-Annual General Meeting Update

Halifax, Nova Scotia, 4 February, 2022 – Today, MedMira Inc. (MedMira) (TSXV: MIR) provides a pre-Annual General Meeting (AGM) update on its regulatory and product development progress. This includes its COVID-19 and its infectious disease products with the latest pre-submission for MedMira’s HCV Rapid Test to the FDA. This update serves the purpose to inform […]
Update on Validation of VYRA™ CoV2Flu

By analyzing the mutation sites of N protein and their proportion according to the coronavirus sequences released by the GISAID database, full-length N protein recombinant antigens corresponding to VOC(Variants of Concern) and other mutant strains have been obtained as a mean to validate the current VYRA™ CoV2Flu antigen test’s sensitivity. The testing variants includes: Omicron […]
MedMira Announces VYRA™ CoV2Flu and Additional Regulatory Opportunities

Halifax, Nova Scotia, 21 December, 2021 – Today, MedMira Inc. (MedMira) (TSXV: MIR) announces the expansion of its VYRATMproduct line by launching its VYRA™ CoV2Flu Rapid Antigen Rapid Test (VYRA™ CoV2Flu). Based on MedMira’s proprietary technology, this 3-in-1 test is developed to distinguish between SARS-CoV-2 and Influenza A or B infections. Furthermore, the Company takes […]



















